Arginine-vasopressin (AVP) is known as the antidiuretic hormone, and it regulates a vital function of our body, water homeostasis. It plays a role in the kidney at the basolateral membranes of the principal cells of the distal collecting duct of the nephron where it directly interacts with the V2 receptor subtype (V2R), which is a typical G protein-coupled receptor (RCPGs). Upon interaction, AVP activates a signaling pathway leading to fusion of aquaporine water channels to the apical membrane of the cells and consequently to water reabsorption from urine to blood. Coupling of the V2R to the Gs protein constitutes a first key step of the cellular response.
But how does it work at a molecular level?
This is what was very recently determined by the team of Bernard Mouillac and Sébastien Granier at the Institut de Génomique Fonctionnelle (IGF), in collaboration with the team of Patrick Bron from the Centre de Biologie Structurale (CBS) using a cryo-electron microscopy approach. The strategy of the researchers also combines experimental NMR (Nuclear Magnetic Resonance) data obtained by Hélène Déméné (CBS) and computational molecular dynamic simulations developed by Nicolas Floquet at the Institut des Biomolécules Max Mousseron (IBMM). This work is 100% done in Montpellier.
This structural biology hybrid approach allowed to solve molecular details of AVP binding to V2R and of the interface of the receptor with the Gs protein signaling partner. Interestingly, the 3D structure of the AVP-V2R-Gs ternary complex had never been defined from an experimental point of view, and represents a real breakthrough.
This study goes further than a simple description of a receptor or a signaling protein complex structure. Indeed, many single mutations of the V2R are responsible for two rare genetic diseases with opposite clinical outcomes: 1/ the congenital Nephrogenic Diabetes Insipidus (cNDI) associated to loss-of-function mutations leading people suffering for this pathology to be unable to concentrate their urine, 2/ the Nephrogenic Syndrome of Inappropriate antidiuresis (NSIAD) linked to constitutive active mutations and characterized by water loading and hyponatremia. V2R is also a major therapeutic target for treating autosomal dominant polycystic kidney disease, a much more frequent pathology which leads in most of the cases to renal failure. Towards these different diseases, there is a real interest in the development of novel V2R « drugs » displaying a better selectivity, a better potency, with no adverse effects.
In particular, the study gives structural hypotheses that may explain consequences of several V2R mutations. For instance, the researchers propose an explanation to link mutants of the amino-acid residue arginine 137 into leucine or cysteine (R137L or R137C) and constitutive activity responsible for NSIAD. It is also tempting to interpret cNDI clinical observations or in vitro pharmacological and cellular data based on the present structure of the V2R. For instance, hypotheses are proposed to explain structural consequences of mutations V88M (valine mutated into methionine) or M272R (methionine mutated into arginine) that are responsible for V2R loss-of-function.
In the future, the knowledge of molecular details of the different conformations of the V2 receptor will guide development of new therapeutic molecules able to activate or to inhibit its function. This research perspective is crucial regarding patients who are suffering for these pathologies which are difficult to manage.
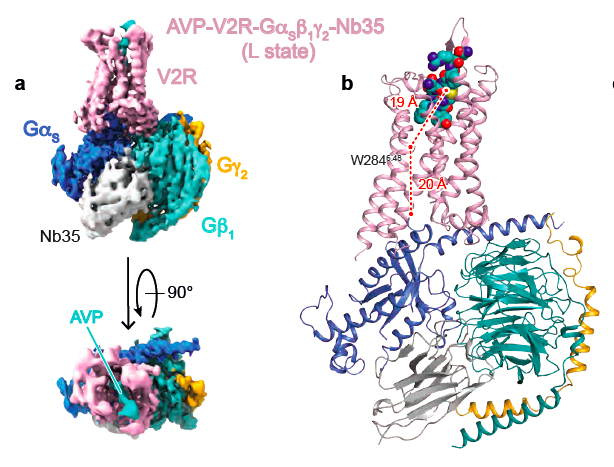
Density map of the AVP-V2R-Gs signaling complex (A) and its corresponding 3D model (B)


