Many diseases of the central nervous system, such as autism, Parkinson’s disease, schizophrenia, anxiety and depression, are linked to the dysfunction of neurotransmitters and their receptors. The development of drugs or therapies against these dysfunctions requires a better molecular and chemical understanding of the activation mechanisms of neurotransmitter receptors. This has been achieved for one of the receptors for glutamate, the main excitatory neurotransmitter in the brain, thanks to a collaboration between France, India and England. These results, published in the journal Cell Reports, have revealed certain molecular structures that control the transmission of the signal between neurons.
The human brain is composed of billions of neurons interconnected by synapses. The neural communication networks thus formed are fundamental to brain functions such as learning processes or memory. Synapses are the site of exchange of communication molecules between neurons, also known as neurotransmitters, such as glutamate. Glutamate, released in the synapse, is taken up by membrane receptors that participate in the modulation of neuronal activity. Among these, the metabotropic subtype 5 glutamate receptor, mGlu5, plays a key role in synaptic function. As its dysfunction is associated with several pathologies of the central nervous system, such as autism, chronic pain, Parkinson’s disease and schizophrenia, understanding its activation mechanisms at the atomic level is essential for developing new drugs to treat these diseases.
In collaboration with a team from the National Centre for Biological Sciences, Tate Institute of Fundamental Research (NCBS-TIFR, Bangalore, India), the Medical Research Council Laboratory of Molecular Biology (MRC-LMB, Cambridge, England), the Paul Scherrer Institute in Switzerland (PSI, Villigen, Switzerland), and the Laboratory of Medicinal Chemistry and Synthesis (MCS, Barcelona, Spain), Guillaume Lebon’s team from the Institute of Functional Genomics (CNRS/INSERM/University of Montpellier) has solved the three-dimensional structure of the mGlu5 receptor linked to chemical agents modulating its activity.
Using microscopy under cryogenic conditions on microscopes at the LMB in Cambridge, they studied the diversity of conformational changes that allow the transmission of the chemical message by the mGlu5 receptor, a phenomenon called signal transduction. They then broke down the molecular functioning of this receptor and of its membrane domain in even greater detail by combining X-ray crystallography with the use of a light-sensitive modulator that allows the activity of the receptor to be controlled with light. These three-dimensional structures and the functional analysis carried out in collaboration between researchers from the IGF, the Centre for Structural Biochemistry (CBS) and the Max Mousseron Institute of Biomolecules (IBMM) in Montpellier have made it possible to highlight the structural and molecular bases that control the equilibrium of the conformations responsible for signal transduction. These results were published in the journal Cell Reports and should help to combat the central nervous system pathologies in which the receptor is involved.
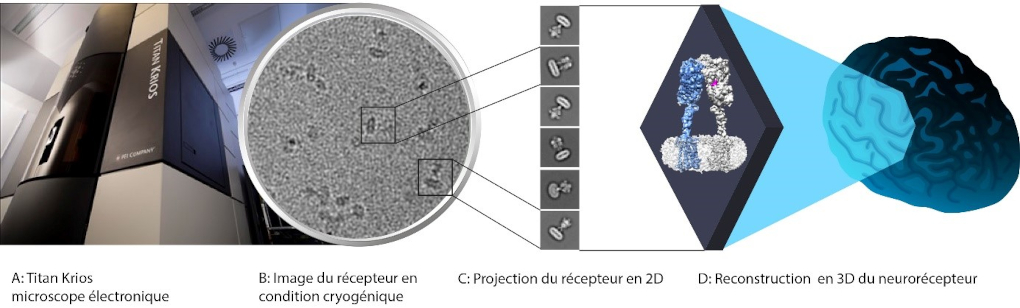
3D structure of the human metabotropic glutamate subtype 5 neuroreceptor, mGlu5, obtained by cryo-electron microscopy. © Guillaume Lebon


