Team Philippe RONDARD
Neuroreceptors, dynamics and functions
Project GPCR complexes
PRINCIPAL INVESTIGATOR

IGF staff involved
Élise JACQUIER
Post-doc CNRS
Imane MAGHNI
IR CNRS
David LEQUEN
IE CNRS
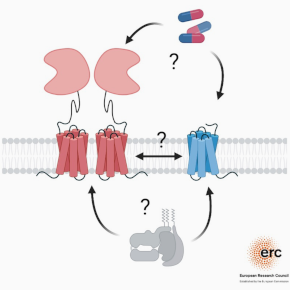
G protein-coupled receptors typically function as individual signaling units at the membrane but can also interact with other GPCRs to form what is called “heteromers”. These heteromers have functional properties that are different from that of the individual GPCRs thus significantly increasing a cell’s capacity to sense and react to its environment.
Consequently, they form new pharmaceutical targets for designing therapeutics with improved tissue-specificity and less side effects. For example, targeting the heteromer AT1-CB1 in the treatment of liver fibrosis, or heteromer mGlu2-5HT2A for schizophrenia.
Our project aims at understanding how heteromers are formed and how each receptor functionally modulates the other in these dynamic assemblies. We organize our research around two main axes:
1/ What is the role the receptor’s environment play in forming GPCR heteromers? Specifically, how lipids and the membrane environment structurally and functionally influence individual receptors and their ability to form larger complexes.
2/ What are the molecular mechanisms of heteromer assembly and of the structural and functional regulations within the complexes?
To answer these questions, we employ different biochemical and biophysical approaches, such as:
- Homogenous Time-Resolved FRET in the form of functional and conformational sensors, as well as lab-developed nanobodies to track the behaviors of receptors in different systems.
- Reconstitution in various model membrane systems to pinpoint the exact role lipids plays in these assemblies.
- Single-particle cryoEM to solve high resolution structures of receptors and their complexes.
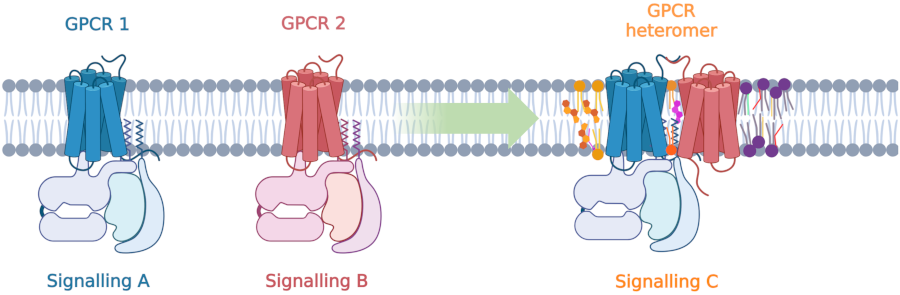
GPCRs can function as individual signaling units at the membrane and can also assemble in complexes termed heteromer which have distinct signaling outputs. Formation of these heteromers could be influenced by the membrane composition.
Main publications
• Menny A., Lukassen MV., et al. Nat Commun, 2021. doi: 10.1038/s41467-021-26366-w
• Lefebvre SN., et al. eLife 2021;10:e60682
• McMahon O., et al. Human Molecular Genetics, 2021. doi: 10.1093/hmg/ddab086.
• Parsons ES., et al. Nat Commun, 2019. doi: 10.1038/s41467-019-10058-7.
• Menny A., et al. Nat Commun, 2018. doi: 10.1038/s41467-018-07653-5.
• Hu H., et al. Proc Natl Acad Sci U S A. 2018. doi: 10.1073/pnas.1813378116.
• Menny a., et al. eLife, 2017. doi: 10.7554/eLife.23955.
• Dumoux M., et al. J Cell Sci, 2015. doi: 10.1242/jcs.169318.
• Sauguet L., et al. Proc Natl Acad Sci U S A., 2014. doi: 10.1073/pnas.1314997111.
Funding
• 2024-2029 ERC Starting Grant, SignalHet – Coordinator
• 2023-2024 ANR T-ERC, SignalHet – Coordinator
• 2021-2025 ANR JCJC, Schizoceptor – Coordinator
Collaborations
• Emmanuel Margeat & Robert Quast (Montpellier)
Alumni
• David Lequen (Master 2)
• Justine Dansette (Master 1)


