Team François RASSENDREN
Purinergic signaling and brain inflammation
Project Roles of Clec7a in Alzheimer’s disease
PRINCIPAL INVESTIGATOR

IGF staff involved
Matthieu PRIEUR
PhD student UM
Coralie CLUA-PROVOST
PhD student UM
Valentin GARCIA
PhD student Inserm
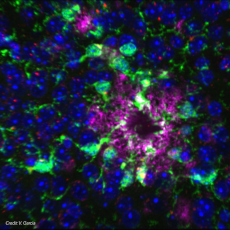
Microglial cells are recognized as major players in brain function. Highly involved in monitoring the nervous parenchyma, they play a key role in triggering neuroinflammatory processes. Equipped with a vast array of receptors and transmembrane proteins, microglia are able to detect changes in the tissue environment and initiate intracellular signals that ultimately lead to a microglial reaction.
Our research group focuses on understanding the mechanisms of microglial reaction initiation, particularly in the context of Alzheimer’s disease.
Functional roles of Clec7a in early Alzheimer’s disease
Human microglial cell diversity in Alzheimer’s disease
Microglial cell diversity in a primate model of Alzheimer’s disease
Functional roles of Clec7a in early Alzheimer’s disease
Our previous work led us to identify Clec7a as an early marker of microglial reaction in Alzheimer’s disease. Nevertheless, the functional role of this receptor remains largely unknown. Our main objectives are to:
• Identify the functional role of Clec7a in microglial cell function
• Determine whether it could represent an early therapeutic target in the context of Alzheimer’s disease.
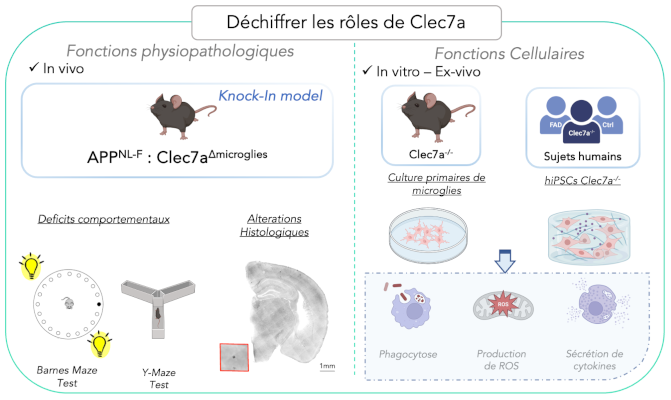
Deciphering the functional roles of Clec7a in early Alzheimer’s disease to determine whether this receptor may represent a therapeutic target.
Main publications
• Hemonnot-Girard AL., et al.; Journal of Neuroinflammation. 2022;19(1):234.
• Hemonnot-Girard AL et al., Brain Behav Immun 2021 91: 404-417.
• Hemonnot AL et al., 2019 Frontiers in Aging Neuroscience.
Funding
• 2023-2025 Fondation Alzheimer – Coordinateur
• 2021-2023 France Alzheimer – Coordinateur
Collaborations
• Dr Carole Escartin (NeuroPsi, Paris-Saclay)
Alumni
• Anne-Laure Hemonnot (Thèse, 2017-2021)
Human microglial cell diversity in Alzheimer’s disease
Although murine and human microglial cells are very similar, there are notable molecular and functional discrepancies, which may explain, in part, the high failure rate of clinical trials, particularly in the context of Alzheimer’s disease.
The aim of the project is to use hiPSC technology to generate immunocompetent neural networks containing neurons, astrocytes, oligodendrocytes and microglia, and to study the molecular and functional heterogeneity of human microglia in the early stages of the disease.
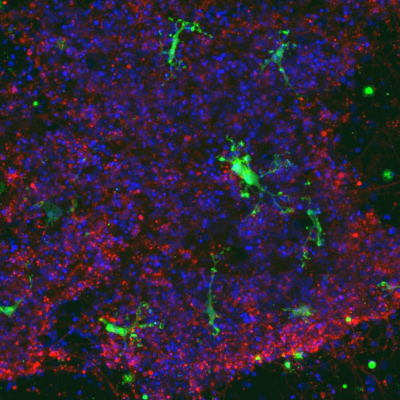
Human iPS-derived microglial cells (IBA1/ green), cocultured with iPS-derived neurons (βIII-tubulin/ red), show a branched morphology, typical of microglia in vivo. Image acquired with the Opera Phenix confocal microscope. Cell nuclei stained with DAPI (blue). Credit C. Clua-Provost/ Carole Crozet.
Main publications
• Clua Provost C. et al. ; Stem Cell Res. 2023 73:103250.
Funding
• 2018-2021 Fondation Alzheimer – Coordinateur
• 2019 Prix de la Fondation Claude Pompidou – Partenaire
Collaborations
• Dr Carole Crozet (INM, Montpellier)
Microglial cell diversity in a primate model of Alzheimer’s disease
To overcome the inherent limitations of genetically distant mouse models and inherently reductionist cell models, a preferred approach is to use non-human primates. Dr. M. Dhenain and his team have developed a primate model by inoculating the microcebe with extracts from patients’ brains, thus reproducing many features of the disease. Through this collaboration, we aim to characterize the diversity of microglial states in this model and analyze their evolution during the progression of the pathology.
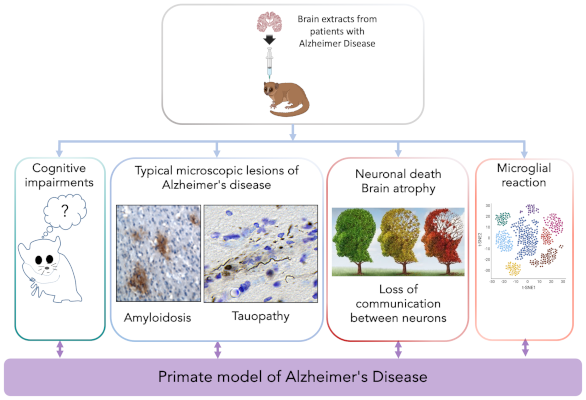
The “Microcebe” model of Alzheimer’s disease, an innovative model to decipher the importance of the microglial response in this pathology.
Funding
• 2022-2026 ANR PrimAlz – Partenaire
Collaborations
• Dr Marc Dhenain (Laboratoire des maladies neurodégénératives, Kremlin-Bicêtre)


