Team Julie PERROY
Pathophysiology of synaptic transmission
Project Dynamics of the mGlu5 receptosome: implications for synaptic transmission and plasticity
PRINCIPAL INVESTIGATOR

IGF staff involved
Nathalie BOUQUIER
IE CNRS
Aragorn VELEZ
Doctorant CNRS
Pierre VINCENT
DR2 CNRS
Yan CHASTAGNIER
Doctorant UM
Julie PERROY
DR1 CNRS
Jeremie NAUDE
CRCN CNRS
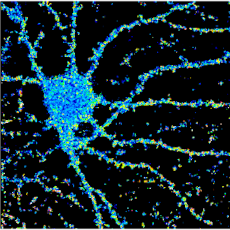
Our work shows that neuronal activity regulates the molecular dynamics of interactions within the glutamatergic receptosome. In turn, this dynamic specifies receptor signaling, modulates synaptic transmission and underlies long-term plasticity mechanisms. We can therefore assume that in each spine, depending on its protein content, a given signal will lead to a particular remodeling of the receptosome. We are studying the dynamics and influence of each of these interactions on receptor function and signalling, synaptic transmission and plasticity mechanisms. We are also working on cell models (including neurons derived from patient hiPSCs) and mouse models of brain pathologies in which the molecular dynamics are disrupted, seeking to repair the deficient interaction in order to regulate neurotransmission finely and transiently.
Synaptic plasticity depends on the strengthening of specific synaptic connections between neurons. But why are some synaptic contacts strengthened and not others that have received the same signal? The synaptic labeling and capture hypothesis proposes that synapses activated during the early phase of plasticity are labeled, enabling them to capture the proteins needed to maintain long-term plasticity. Taken together, these mechanisms would trigger a synapse in a functional network. Our preliminary results suggest that protein dynamics of the mGlu5 receptosome may enable a spine to be selected and maintained in a potentiated state. We are testing this hypothesis using mainly BRET approaches to study the interaction dynamics of proteins of interest in real time, at the spine scale. We assess plasticity using super-resolution microscopy approaches by measuring dendritic spine volume and membrane insertion of AMPA receptors. As proof of concept of the importance of these molecular mechanisms in cognition, we are also seeking to repair deficient protein interactions in mouse models of Autism Spectrum Disorder.
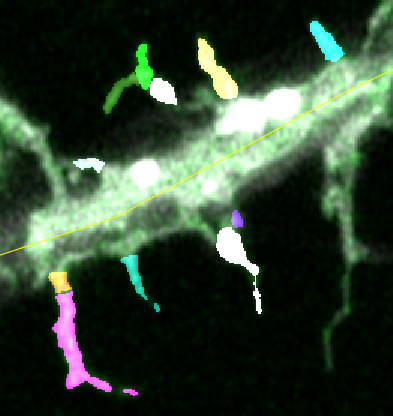
Identification of spines using super-resolution imaging, with the aim of measuring their changes in volume and the membrane insertion of AMPA receptors during plasticity.
Main publications
• Moutin E., et al. (2021) Molecular Psychiatry. doi: 10.1038/s41380-021-01230-x
• Bouquier N., et al. (2020) BMC biology doi:10.1186/s12915-020-00790-8
• Moutin E., et al. (2016) Cerebral cortex. doi: 10.1093/cercor/bhw183
• Moutin E., et al. (2012) J Cell Biol, 2012. doi:10.1083/jcb.201110101
Funding
• Soutien à la recherche 2024 – programme d’excellence I-SITE
• ANR LEARN 2022
• ANR ISIBRAIN 2022
Collaborations
• Carlo Sala (Italy)
• Chiara Verpelli (Italy)


