Team Julie PERROY
Pathophysiology of synaptic transmission

Through their ability to form and modify their synaptic connections, our neurons are constantly remodelled by our experiences, which modifies our functional neural networks and our interaction with the environment. Our work aims to understand how learning emerges from plasticity, using a multi-scale approach, from the dynamics of synaptic molecules to the cellular and behavioral events that do or do not trigger plasticity. Understanding this plasticity will not only lead to the discovery of new therapies for neurological diseases and psychiatric disorders, but will also provide answers to fundamental questions about cognition.
We are investigating the molecular and cellular determinants that regulate the formation, function and plasticity of synapses, as well as the signalling of ensembles of neurons responsible for learning behavior. This enables us to approach the question of behavioral disorders, whether they result from genetic or environmental factors, from the angle of molecular, cellular and neuronal network mechanisms.
Neuronal plasticity is a constantly active process: neuronal activity modifies synaptic properties, which in turn change the activity of neuronal networks. We are therefore focusing on the dynamics of synaptic proteins involved in glutamate receptor subcellular trafficking, membrane targetting and signalling. Our previous work show that the different functions of a receptor rely on its ability to engage in specific protein-protein interactions with scaffolding and effector proteins to form a functional unit called a receptosome. These protein-protein interactions are finely regulated, in a highly dynamic way, by interaction with the environment, and therefore need to be studied from a systems biology perspective, combining predictions from theoretical models and cutting-edge experimentation. For example, understanding whether a receptor is activated as a free molecule or as part of different complexes and what its causal role is in each of these conditions are key biological issues in understanding the fundamental logic of plasticity. This knowledge also offers the possibility of exclusively targeting the altered signalling pathway in a given pathology.
Our projects aim to characterise the dynamics of synaptic molecules, the logic of the signalling and plasticity system of a neuron (and ensembles of neurons), and the resulting behavioral learning. We work within a formal theoretical framework (biophysical and cognitive modelling) that links these scales. On the one hand, this approach provides an explanatory framework describing how properties at one level of organisation (e.g. neural networks) emerge from lower levels (interactions between neurons, between molecules); on the other hand, it provides quantitative predictions (what form of plasticity, what molecule involved) to guide our experiments, which then answer specific questions. In order to characterise these different scales of organisation (molecules, neuron, network, behavior), we are developing methods for the real-time study of protein-protein interaction dynamics and neuronal signalling in vitro and in vivo, as well as tools for constraining molecular dynamics at synapse level and understanding their physiological and therapeutic importance. Finally, by taking advantage of cell and mouse models, we are promoting mechanistic translational research, going back and forth from the mouse to the patient. To propose therapeutic targets, we can reproduce a genetic mutation identified in a patient with neurodevelopmental disorders and/or expose the mice to an environmental factor, in order to overcome the correlations between factors and disorders and understand their causality.
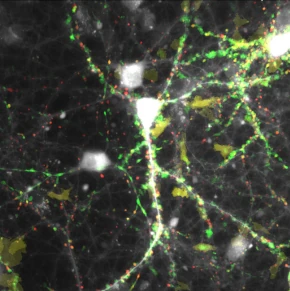
Projection of calcium events recorded over 2 minutes with GCamP6s in hippocampal neurons, sorted according to size and spread

 IGF Sud 227
IGF Sud 227 04 34 35 92 10
04 34 35 92 10
 IGF Sud 227
IGF Sud 227 04 34 35 92 10
04 34 35 92 10
 IGF Sud 227
IGF Sud 227 04 34 35 92 10
04 34 35 92 10
 IGF Sud 227
IGF Sud 227 04 34 35 92 10
04 34 35 92 10
 IGF Sud 227
IGF Sud 227 04 34 35 92 10
04 34 35 92 10
 IGF Sud 227
IGF Sud 227 04 34 35 92 10
04 34 35 92 10
 IGF Nord 223
IGF Nord 223 04 34 35 92 10
04 34 35 92 10
 IGF Nord 223
IGF Nord 223 04 34 35 92 10
04 34 35 92 10
 IGF Sud 227
IGF Sud 227 04 34 35 92 10
04 34 35 92 10
 IGF Sud 227
IGF Sud 227 04 34 35 92 10
04 34 35 92 10
 IGF Sud 227
IGF Sud 227 04 34 35 92 10
04 34 35 92 10
 IGF Nord 223
IGF Nord 223 04 34 35 92 12
04 34 35 92 12
 IGF Sud 227
IGF Sud 227 04 34 35 92 10
04 34 35 92 10
 IGF Sud 227
IGF Sud 227 04 34 35 92 10
04 34 35 92 10
 IGF Sud 227
IGF Sud 227 04 34 35 92 12
04 34 35 92 12
 IGF Sud 227
IGF Sud 227 04 34 35 92 10
04 34 35 92 10
 IGF Sud 227
IGF Sud 227 04 34 35 92 10
04 34 35 92 10
 IGF Sud 227
IGF Sud 227 04 34 35 92 10
04 34 35 92 10
 IGF Sud 227
IGF Sud 227 04 34 35 92 10
04 34 35 92 10
 IGF Sud 227
IGF Sud 227 04 34 35 92 10
04 34 35 92 10ALUMNI
• Sophie Sakkaki – Préparation de l’Agrégation en SVT
• Noémie Cresto – Chercheuse en Neurosciences (Post-doc, IGF Montpellier, France)
• Benoît Girard – Chercheur en Neurosciences (Université de Genève, Suisse)
• Marin Boutonnet – Chercheur en Neurosciences (Post-doc, Humboldt Université de Berlin, Allemagne)
• Roza Szlendak – Chercheuse en Neurosciences (IMC, warsaw, Pologne)
• Federica Giona – Spécialiste recherche et développement (recherche d’emploi, Milan Italie)
• Federica Bertaso – Chercheuse en Neurosciences (CRCN INSERM – IGF Montpellier, France)
• Jeanne Ster – Chercheuse en Neurosciences (CRCN CNRS – IGF Montpellier, France)
• Elise Goyet – Educatrice spécialisée (Lyon, France)
• Fabrice Raynaud – Enseignant chercheur (Université de Montpellier, France)
• Laura Ceolin – Attachée de recherche Clinique (Mediaxe Formation, Paris, France)
• Selma Dadak – Chercheuse en Neurosciences (Sensorion, Montpellier, France)
• Valériane Tassin – Webmaster (France)
• Adeline Burguière – Enseignante cours particuliers (Matha2, Montpellier, France)
• Jonathan Roger – Responsable des opérations cliniques (Paraxel, Montreal, Quebec, Canada)
- Boutonnet, M., Carpena, C., Bouquier, N., Chastagnier, Y., et al. (2024). Voltage tunes mGlu5 receptor function, impacting synaptic transmission. J. of Pharmacol. – in press
- Sakkaki, S., Cresto, N., Chancel, R., Jaulmes, M., Zub, E., Blaquière, M., et al. (2023). Dual-Hit: Glyphosate exposure at NOAEL level negatively impacts birth and glia-behavioural measures in heterozygous shank3 mutants. Environ Int doi: 10.1016/j.envint.2023.108201
- Areias, J., Sola, C., Chastagnier, Y., Pico, J., Bouquier, N., Dadure, C., et al. (2023). Whole-brain characterization of apoptosis after sevoflurane anesthesia reveals neuronal cell death patterns in the mouse neonatal neocortex. Sci Rep doi: 10.1038/s41598-023-41750-w
- Bousseyrol, E., Didienne, S., Takillah, S., Prevost-Solié, C., Come, M., Ahmed Yahia, T., et al. (2023). Dopaminergic and prefrontal dynamics co-determine mouse decisions in a spatial gambling task. Cell Rep doi: 10.1016/j.celrep.2023.112523
- Moutin, E., Sakkaki, S., Compan, V., Bouquier, N., Giona, F., Areias, J., et al. (2021). Restoring glutamate receptosome dynamics at synapses rescues autism-like deficits in Shank3-deficient mice. Mol Psychiatry. doi: 10.1038/s41380-021-01230-x
- Sarazin, M. X. B., Victor, J., Medernach, D., Naudé, J., and Delord, B. (2021). Online Learning and Memory of Neural Trajectory Replays for Prefrontal Persistent and Dynamic Representations in the Irregular Asynchronous State. Front Neural Circuits doi: 10.3389/fncir.2021.648538
- Mota, É., Bompierre, S., Betolngar, D., Castro, L. R. V., & Vincent, P. (2021). Pivotal role of phosphodiesterase 10A in the integration of dopamine signals in mice striatal D1 and D2 medium-sized spiny neurones. British Journal of Pharmacology, 178(24). doi: 10.1111/bph.15664
- Bouquier, N., Moutin, E., Tintignac, L., Reverbel, A., Jublanc, E., Sinnreich, M., et al. (2020). AIMTOR, a BRET biosensor for live imaging, reveals subcellular mTOR signaling and dysfunctions. BMC Biol. Jul 3;18(1.
- Sebastianutto, I., Goyet, E., Andreoli, L., Font-Ingles, J., Moreno-Delgado, D., Bouquier, N., et al. (2020). D1-mGlu5 heteromers mediate noncanonical dopamine signaling in Parkinson’s disease. Journal of Clinical Investigation 130, 1168–1184. doi: 10.1172/JCI126361
- Nair, A. G., Castro, L. R. V., El Khoury, M., Gorgievski, V., Giros, B., et al. (2019). The high efficacy of muscarinic M4 receptor in D1 medium spiny neurons reverses striatal hyperdopaminergia. Neuropharmacology (2019) 1. doi: 10.1016/j.neuropharm.2018.11.029
Dynamics of the mGlu5 receptosome: implications for synaptic transmission and plasticity
Principal investigator
Enora MOUTIN

Cellular and mouse models enable a better understanding of the molecular and cellular mechanisms behind behavioral disorders and to test new therapeutic approaches. Several clinical situations are being studied in the laboratory.
- Autism spectrum disorders (ASD): Neurodevelopmental and/or cognitive disorders are caused by mutations in genes coding for synaptic proteins, such as glutamate receptors (GRIN2B), scaffolding proteins that control their function (SHANK3) or factors regulating gene expression during plasticity (FMR1). We are also studying the impact of environmental factors on brain function, factors as varied as exposure to pesticides (Glyphosate), viruses (SARS-CoV-2) or stress.
- Subarachnoid haemorrhage: Subarachnoid hemorrhage by aneurysm rupture is often followed by delayed cerebral ischemia, notably grieving patients prognosis. Pathophysiology of this complication is currently unclear. We aim at exploring, in an experimental model, the microvascular defects and the microglial response to subarachnoid hemorrhage using in vivo microscopy, MRI, tissue clearing and transcriptomics. In a translational approach, we also study markers of these phenomena in patients using MRI and dynamic PET imaging.
- General anaesthetic agents: General anesthesia triggers drastic changes in neuronal activity throughout the entire brain. This may be accompanied by mid/long-term changes in cellular functions. We aim to study a few signaling pathways of interst using tissue clearing methods and immunohistochemistry. Relevant identified structures are then characterized using a panel of approaches including in vivo extracellular electrophysiological recordings.
Funding
• ANR (2023-2027) LEARN “Light on intracellular signaling Events shAping functional netwoRks in spatial mapping”
• ANR (2023-2027) IsiBRAIN “Impact of SARS-CoV-2 Infection on the central nervous system”
• ANR (2024-2028) TimeTag “Timed decisions: underlying neural mechanisms and TAGging of neuronal ensembles in the prefrontal cortex”
• ANR (2020-2024) Glyflore “Impact of a chronic dietary exposure to low-doses of glyphosate on the gut microbiota and microbiota-associated physiological functions”
• CCA-Inserm-Bettencourt (2022-2026) “Role of microcirculatory damage in delayed cerebral ischaemia complicating aneurysmal subarachnoid haemorrhage”
• FRC (2024-2026) “Multiscale scientific method to fully characterize the functional consequences of GRIN variants, from biological mechanisms to brain diseases”
• FRM (2023-2026) “Role of mGlu5 receptosome dynamics in plasticity, metaplasticity and flexibility – Alleviation of autism spectrum disorders by molecular tinkering”
• MUSE (2020-2024) “Environmental pesticides and genetic predisposition: a path toward autism spectrum disorders “
• MUSE (2024) STRESSA “Combined effects of environmental stress and genetic predisposition to Autism Spectrum Disorders”
Collaborations
 • Cathie Ventalon (IBENS, Paris France)
• Cathie Ventalon (IBENS, Paris France)
• Raphael Gaudin (IRIM, Montpellier France)
• Stéphanie Trouche (IGF, Montpellier France)
• Bruno Delord (ISIR, Paris France)
• Nicola Marchi et Etienne Audinat (IGF, Montpellier France)
• Sandrine Ellero-Simatos et Laurence Payrastre (INRAe, Toulouse France)
• Dorota Hoffman (IMC, Warsaw, Poland)
• Chiara Verpeli et Carlo Sala (CNS, Milano Italie)
• Amaria Baghdadli (CRA, CHU Montpellier France)
• Hélène Hirbec (IGF, Montpellier France)
• Noura Faraj (LIRMM, Montpellier France)
• Christophe Goze-Bac (L2C, Montpellier France)
• Bertrand Wattrisse (LMGC, Montpellier France)
• Emmanuelle Le Bars (I2FH, Montpellier France)
• Denis Mariano-Goulart (CHU, Montpellier France)
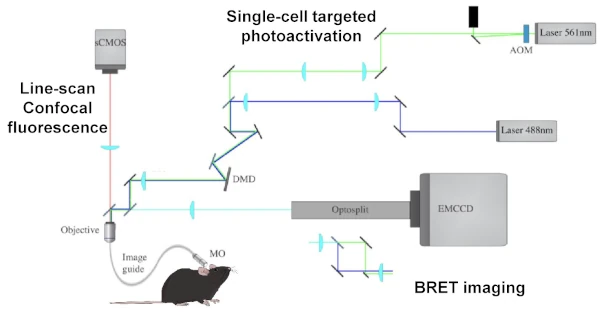
The tools and methods currently available limit our ability to study the dynamics and function of protein-protein interactions and neuronal signalling. To overcome these technological limitations, we are using and developing :
- biosensors for signalling pathways involved in neuronal plasticity (Fluorescence, BRET, FRET), with the ambition of combining readings from several signalling pathways (multiplexing), simultaneously.
- actuators to force, prevent or repair synaptic molecular dynamics and understand their physiological and therapeutic relevance.
- Plugins for automating image acquisition and analysis, to resolve the complexity of a single signal and the intricacies of multiple stimuli.
- new technologies, such as BRET imaging in living cells for subcellular localisation of protein dynamics in real time, as well as in vivo BRET for recording the signalling of neuronal ensembles in animals during behavior.