Team Philippe MARIN
Neuroproteomics and signalling of brain disorders
Project Microbiota, serotonin and neurodegenerative diseases
PRINCIPAL INVESTIGATOR

IGF staff involved
Philippe MARIN
DRCE, CNRS
Joël BOCKAERT
PU, UM
Marie-Laure PARMENTIER
DR2, Inserm
Sylvaine ARTERO
CRCN, Inserm
Lucy KUNDURA
Post-doc, Inserm
Thomas CUREL
PhD student, Inserm
Sonya GALANT
IECN, CNRS
Based on expertise in the neuropharmacology of G-protein receptors (GPCRs), in particular serotonin (5-HT) receptors, and pioneering work on gut microbiota, this research group aims to propose innovative therapies for neurodegenerative diseases, in particular Alzheimer’s disease. This goal can be divided into 3 areas:
5-HT4 receptor proteome and prevention of Alzheimer’s disease
5-HT4 receptor proteome and prevention of Alzheimer’s disease
Our goal is to develop a new therapeutic approach aimed at increasing APP cleavage by the a-secretase ADAM10 to counteract APP maturation towards the non-amyloidogenic pathway.
Our main tool is the serotonin type 4 receptor (5-HT4), which allows us to:
- Promote intracellular trafficking of the a-secretase ADAM10 to the plasma membrane, where it is active
- Stimulate ADAM10 and the production of the soluble fragment sAPPa with neuroprotective properties
- Use proteomics to search for new protein partners that affect APP trafficking and/or metabolism.
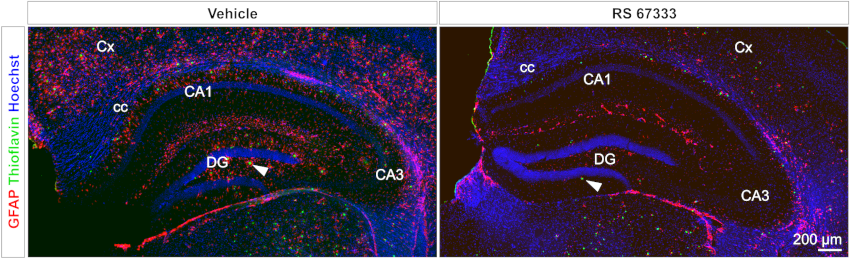
Reduction of amyloidosis and astrogliosis by activation of the 5-HT4 receptor.
5xFAD mouse model treated with RS 67333 (5-HT4R agonist). Amyloid plaques (Thioflavine T, green). Astrocytes (GFAP, red). From Giannoni P. Front Aging Neurosci. 2013.
Main publications
• Giannoni P., et al. (2020) Front Aging Neurosci, 12, 3.
• Baranger K., et al. (2017) Neuropharmacology, 126, 128-141.
• Giannoni P., et al. (2016) Neurobiol Dis, 88, 107-17.
• Arango-Lievano M., et al. (2016) J Vis Exp, 6, 54796.
• Saraceno C., et al. (2014) Cell Death Dis, 5, e1547.
• Giannoni P., et al. (2013) Front Aging Neurosci, 5, 96.
• Cochet M., et al. (2013) ACS Chem Neurosci, 4, 130-40.
Funding
• 2015-2018 Fédération pour la Recherche sur le Cerveau (FRC 2015) – Partenaire
• 2013-2018 ANR 2012-ADAMGUARD – Responsable de tâche
• 2012-2015 Région Languedoc Roussillon – Investigateur Principal
• 2011-2014 Association France Alzheimer – Investigateur Principal
Collaborations
• Stephan Lichtenthaler (DZNE, Munich)
• Monica Di Lucca (Milan)
• Santiago Rivera (Marseille)
• François Roman (Marseille)
• Nicola Marchi (Montpellier)
• Freddy Jeanneteau (Montpellier)
Alumni
• Hugo Payan (PhD, 2016-2020)
• Patrizia Giannoni (Post-Doc., 2011-2016)
• Florence Gaven (IE, 2003-2015)
• Romain Donneger (PhD, 2010-2014)
• Elena Marcello (Post-Doc., 2011-2012)
• Maud Cochet (PhD, 2007-2011)
Innovative multi-target ligands in Alzheimer’s disease
Our consortium proposes to develop multi-targeted directed ligand (MTDL) therapeutics capable of targeting multiple molecular players involved in the pathogenesis of the disease in order to achieve a combination of symptomatic and procognitive effects and impact on the progression of Alzheimer’s disease.
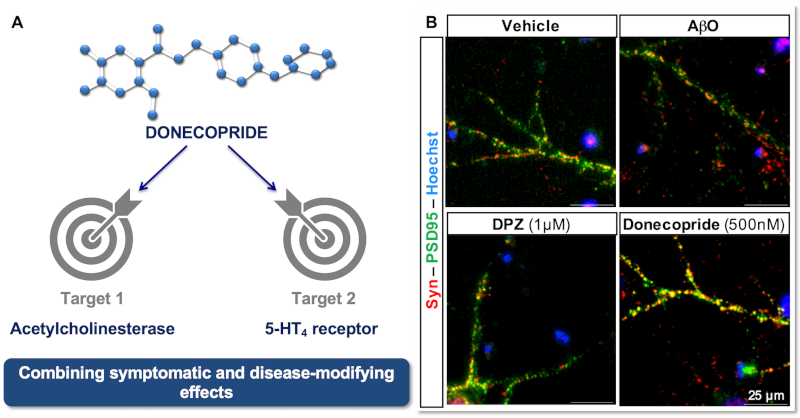
Design and characterisation of donecopride
Drug candidate with both acetycholinesterase inhibitory activity and a 5-HT4 receptor agonist profile leading to a procognitive effect in vivo in a mouse model.
Primary rat hippocampal neurons in the presence of vehicle, oligomers of Ab (AbO), donepezil (DPZ) or donecopride. Synaptophysin (red), PSD95 (green), Hoechst (blue). From Rochais C. Br J Pharmacol, 2020.
Main publications
• Antonijevic M., et al. (2024) Molecules, 29, 515.
• Toublet F. X., et al. (2021) Eur J Med Chem. 210, 113059.
• Lalut J., et al. (2020) Sci Rep, 10, 314.
• Toublet F. X., et al. (2019) Molecules, 24.
• Rochais C., et al. (2020) Br J Pharmacol, 177, 1988-2005
• Lanthier C., et al. (2019) Eur J Med Chem, 182, 111596.
• Hatat B., et al. (2019) Front Aging Neurosci. 11,148.
• Rochais C., et al. (2015) J Med Chem, 58, 3172-87.
• Lecoutey C., et al. (2014) PNAS, 111, E3825-30.
Funding
• 2020-2025 ANR 2020-DOWNALZ – Partner
• 2016-2019 Ligue Européenne Contre La Maladie d’Alzheimer (LECMA) – Coordinator
• 2015-2018 Fondation Plan Alzheimer (AAP 2015) – k€100 (k€33) – Partner
• 2012-2014 Ligue Européenne Contre La Maladie d’Alzheimer (LECMA) – Coordinator
Collaborations
• Patrick Dallemagne (Caen)
• Michel Boulouard (Caen)
• Santiago Rivera (Marseille)
Alumni
• Caroline Ismeurt (Thèse, 2017-2021)
• Bérénice Hatat (Thèse, 2016-2019)
• Hugo Payan (Thèse, 2016-2020)
• Patrizia Giannoni (Post-Doc., 2011-2016)
• Florence Gaven (IE, 2003-2015)
Patents
• Dallemagne P., Rochais C. & Claeysen S. EP 3628660, 2020
• Dallemagne P., Rochais C. & Claeysen S. EP 3628315, 2020
• Dallemagne P., Rochais C. & Claeysen S. WO 2020065028, 2020
• Dallemagne P., Rochais C. & Claeysen S. WO 2020064979, 2020
Gut microbiota and Alzheimer’s disease
Dysbiosis of the gut microbiota is thought to play a central role in the chronic inflammatory process involved in the progression of Alzheimer’s disease. The production of metabolites by certain bacteria is thought to influence peripheral immune cells and promote systemic and central inflammation.
Our aim is to determine the impact of the gut microbiota on the development of Alzheimer’s disease in order to propose biomarkers of bacterial origin and innovative intervention strategies based on restoring the composition of the gut microbiota.

The MICMALZ clinical study NCT04841135 aims to:
- Characterize the blood and gut bacterial signature of Alzheimer’s patients and search for microbiota-derived biomarkers.
- Define the influence of gut dysbiosis in Alzheimer’s patients on cognition and disease progression using microbiota transfer in a mouse model of Alzheimer’s disease.

The DENTALCOG project ANR-22-CE36-0003 aims to:
- Analyse the associations between refined carbohydrate consumption, periodontal disease and cognitive processes and the associated changes in brain and plasma biomarkers in two complementary cohorts of the general population.
Validate the epidemiological findings and to decipher the effects of excessive sucrose consumption on periodontitis, cognition and oral microbiota in an animal model (5xFAD mice).
Funding
• 2023-2026 ANR 2022-DENTALCOG– Partner
• 2021-2024 Programme Transversal INSERM MICROBIOTE 2 – Partner
• 2019-2022 Projet MICMALZ, Région Occitanie/FEDER – Coordinator
• 2017-2020 Programme Transversal INSERM MICROBIOTE 1 – Partner
Collaborations
• Audrey Gabelle (CHU, Montpellier)
• Karim Bennys (CHU, Montpellier)
• Germain Busto (CHU, Montpellier)
• Yves Dauvilliers (CHU, Montpellier)
• Sylvain Lehmann (CHU, Montpellier)
• Xavier Collet (CREFRE, Toulouse)
• Damien Rei (NBX Biosciences, Montpellier)
• Sylvaine Artero (IGF, Montpellier)
• Michel Neunlist & Moustapha Cissé (Nantes)
• Samuel Chaffron (Nantes)
• Agnes Lehuen (Cochin, Paris)
• Sylvie Rabot (INRAE, Jouy-en-Josas)
• Christophe Junot (METABOHUB, CEA)
• Société MaaT Pharma (Lyon)
• Société VAIOMER (Labège)
Alumni
• Linda-Nora Mekki (IE, 2020-2022)
• Caroline Ismeurt (Thèse, 2017-2021)
• Patrizia Giannoni (Post-Doc., 2011-2016)
• Florence Gaven (IE, 2003-2015)


