Team Philippe MARIN
Neuroproteomics and signalling of brain disorders
Project Identification and functional analysis of multiple sclerosis biomarkers
PRINCIPAL INVESTIGATOR

IGF staff involved
Manon RIVAL
PhD student, Tutelle
Multiple sclerosis (MS) is an autoimmune disease of the central nervous system (CNS), characterized by patches of demyelination with lymphocytic and macrophagic infiltration. Chronic glial activation in these plaques inhibits remyelination, leading to axonal damage and long-term disability. There are multiple genetic and environmental risk factors, including vitamin D deficiency and EBV infection, but their role in immune system activation remains poorly defined. The analysis of the effects of Vitamin D on the immune system and the search for MS biomarkers in blood and cerebrospinal fluid (CSF) are major challenges in order to better understand the pathophysiology of the disease and develop new therapies aimed at protecting or repairing myelin.
The project focuses on identifying new biomarkers of multiple sclerosis (MS) using proteomics techniques. Analysis of cerebrospinal fluid (CSF) samples comparing MS patients with controls using a powerful immunoprecipitation method to remove the 20 most abundant plasma proteins from CSF has enabled in-depth characterization of the CSF proteome, including low-abundance proteins secreted by central nervous system cells. In parallel, the use of stable isotope labeling in cell culture (SILAC) to study the neuronal secretome has enabled the identification of several markers of neuronal lesions induced during inflammation or apoptosis. This work led to the identification of CHI3L1 and CHI3L2 as validated biomarkers of MS in CSF, with increased expression in MS plaques in the brains of MS patients. In addition, I analyzed the prognostic value of CSF CHI3L1 levels in patients with RIS (clinically isolated radiological syndrome).
Recently, thanks to technological advances in proteomics, our team has identified new MS biomarkers, including syndecan-1 (also known as CD138, a plasma cell marker), validated by parallel reaction mass spectrometry (PRM) (Patent No. 18305630.8, May 22, 2018). Curiously, Syndecan-1 is expressed in oligodendrocyte precursor cells (OPCs) and acts as a receptor for CHI3L1 in several models. Chitinase 3-like protein 1 (CHI3L1) is an MS biomarker secreted by astrocytes, microglial cells and macrophages. It is involved in extracellular matrix remodeling via its association with various membrane proteins, including the CRTH2 receptor and Syndecan-1, another MS biomarker expressed in oligodendrocyte progenitor cells (OPCs) and discriminating MS from other inflammatory pathologies. The team’s preliminary results suggest that in MS, CHI3L1 may affect the differentiation of OPCs into mature oligodendrocytes involved in remyelination.
We aim to investigate the role of the CHI3L1-SDC1 complex in OPC differentiation and remyelination using murine OPC lines and human pluripotent stem cell-derived OPCs (hiPSC-OPCs). This project will determine whether the CHI3L1-SDC1 complex and the signaling pathways it controls constitute targets for a new therapeutic strategy in MS, particularly for progressive forms of the disease.
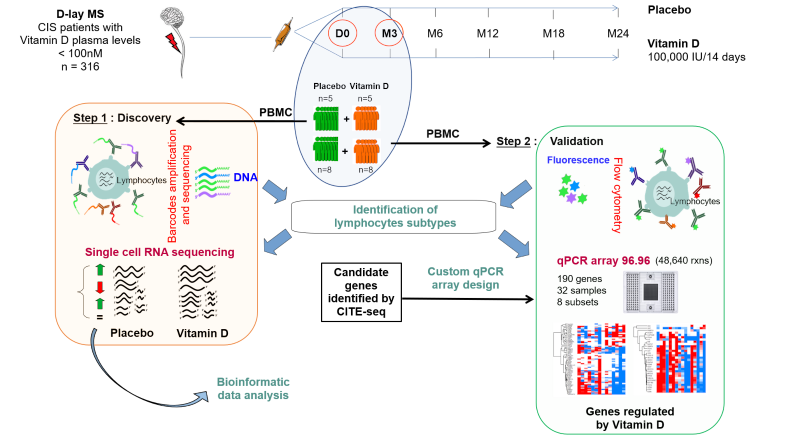
Our research project also focuses on the therapeutic potential of vitamin D in multiple sclerosis (MS). With the results of the D-lay MS clinical trial, the largest double-blind clinical trial to assess the clinical efficacy of vitamin D in reducing conversion to MS after a clinically isolated syndrome, we aim to analyze the immunomodulatory mechanisms of vitamin D in vivo in MS and in healthy subjects. Using a large number of bio-collected samples, we are developing functional analyses of the modulation of lymphocyte activity by vitamin D, and analyzing gene expression changes in circulating lymphocytes from MS patients using single-cell RNA sequencing.
Thanks to a number of collaborations, we now have a mirror-image clinical study in healthy subjects at our disposal, and want to analyze the effects of vitamin D in the human species, not only at lymphocyte level, but also in the intestinal and blood microbiota, as well as in the plasma metabolome. The results of these projects will provide a better understanding of the immunoregulatory effects of vitamin D in the general population, optimize the primary prevention of MS and autoimmune diseases in healthy subjects, and pave the way for new therapeutic targets for the treatment of MS.
Main publications
• Hinsinger, G. et al. (2024) Neurol Neuroimmunol Neuroinflamm 11, e200230
• Rival, M. et al. (2023) Neurol Neuroimmunol Neuroinflamm 10, e200044
• Galoppin, M. et al. (2023) bioRxiv 2023.09.25.559400
• Galoppin, M. et al. (2022) Brain Commun 4, fcac171
• Conejero, I. et al. (2022) Brain 145, 3264-3273
• Rival, M., et al. (2022) Front. Immunol. 13, 866092
• Gauthier, A. et al. (2021) Annals of Clinical and Translational Neurology doi:10.1002/acn3.51355.
• Thouvenot, E. et al. (2020) Eur J Neurol 27, 251-257
• Camu, W. et al. (2019) Neurol Neuroimmunol Neuroinflamm 6, e597
• Thouvenot, E. et al. (2018) Mult. Scler. 1352458518767043
• Prin, P. et al. (2017) Ann. Neurol. 82, 489-490
• Nerrant, E et al. (2016) Neurology 86, e38-39
• Thouvenot, E., et al. (2015) Eur J Neurol 22, 564-569
• Hinsinger, G., et al. (2015) Mult Scler, 21(10):1251-61
• Thouvenot, E. et al. (2012) J Proteomics 77, 394-405
• Rigau, V. et al. (2012) Neurology 79, 2214-2216
• Thouvenot, E. et al. (2008) Journal of Proteome Research 7, 4409-4421
• Thouvenot, E. et al. (2006) Proteomics 6, 5941-52
Funding
• 2024-2027 University of Montpellier / CHU Nîmes – CITE-VDSS – Coordinator
• 2021-2024 National MS Society – ProBioRIS – Partner
• 2020-2021 ARSEP – Coordinator
• 2019-2022 ANR PRCI – VITADIMS – Partner
• 2019-2021 ARSEP – Partner
• 2014-2016 Genzyme- BioCoMS – Coordinator
• 2015-2016 ARSEP – Coordinator
• 2013-2014 ARSEP – Coordinator
• 2012-2015 Université Montpellier – CHU Nîmes – Coordinator
• 2009-2011 ARSEP – Coordinator
• 2008-2009 ARSEP – iTRAQ – Coordinator
• 2005-2007 Inserm host position – PhD
• 2003-2004 FRM – Master 2
Collaborations
• Anne Astier (Toulouse)
• Britta Engelhardt (Bern)
• Catherine Lubetzki (Paris)
• Sylvain Lehmann (Montpellier)
• Sophie Assant (Lyon)
Alumni
• Manon Rival (PhD, 2024-2027)
• Manon Galoppin (PhD, 2020-2023)
• Lucile du Trieu de Terdonck (PhD, 2018-2022)
• Geoffrey Hinsinger (PhD, 2014-2018)
Patents
• Thouvenot E., Marin P., Urbach S., Hinsinger G. EPO 18305630.8, 2018
• Marin P., Thouvenot E., Nguyen G., Groszer M. WO 2013/124406 A1, 2013


