Team Philippe LORY
Ion channels in neuronal excitability and diseases
Project Stimulus-secretion coupling in the adrenal medullary tissue: pathophysiology of the adaptive response to stress and associated disorders
PRINCIPAL INVESTIGATOR

IGF staff involved
Claire BERNAT
IECN CNRS
Arnaud MONTEIL
CRHC CNRS
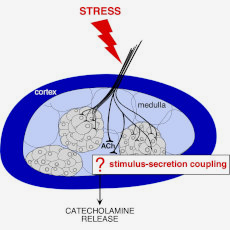
Hormone secretion is a physiological function that is finely regulated to meet the body’s needs. In stressful situations, adrenal chromaffin cells release catecholamines (adrenaline notably), enabling the body to trigger an adaptive response. This secretion is controlled by several cellular and tissue mechanisms, the coordination of which being essential for maintaining the body’s homeostasis. Any change in one of these mechanisms can have an impact on hormone release, with consequences for the body’s physiology.
Plasticity of the adrenal stimulus-secretion coupling in response to arterial hypertension
Influence of the gut microbiota on the stress response of the adrenal medullary tissue
Role of chromaffin cell excitability in the regulation of catecholamine secretion: contribution of sodium leak conductances
In the context of excitability, we are interested in the ion channels operating around the resting membrane potential of cells. In acute slices of mouse adrenal glands, we have identified a conductance permeable to Na+ ions (carried by sodium leakage channels NALCN?), which regulates chromaffin cell electrical activity. The role of NALCN in chromaffin cell excitability and catecholamine secretion was studied in a mouse model genetically manipulated to inactivate NALCN expression in chromaffin cells.
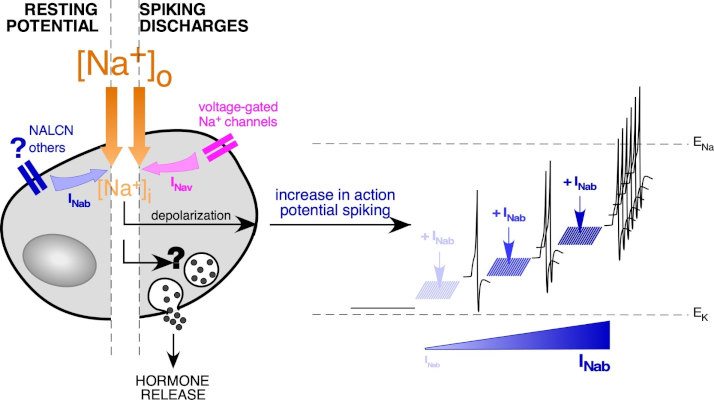
Schematic representation of Na+ homeostasis in a chromaffin cell: focus on ion channel-mediated Na+ entry. The difference in Na+ concentration between the cytoplasm (~10 mM) and the surrounding extracellular medium (~140 mM) is in favor of an inward Na+ flux when Na+-permeant channels open. At the resting membrane potential, the presence of background Na+-permeant channels (NALCN…) generates a persistent Na+ influx (here called INab). The addition of INab, which acts as a depolarizing component, shifts the resting membrane potential value to more positive potentials. This, in turn, increases the spiking discharges. Adapted from Guérineau et al., Front Neuroendocrinol, 2021.
Main publications
• Monteil A., et al. (2024) Physiol Rev, 399, 472.
• Guérineau NC (2023) Methods Mol Biol, 2565, 113-127.
• Milman A., et al. (2021) J Physiol, 599, 1855-1883.
• Guérineau NC., et al. (2021) Front Neuroendocrinol, 63, 100947.
Funding
• 2015-2024 ANR LabEx “Ion Channels, Science and Therapeutics” – Partner
Collaborations
• Pierre Fontanaud (Montpellier)
• Stéphanie Ventéo (Montpellier)
• Jean-Louis Bossu (Strasbourg)
Alumni
• Alexandre Milman (Thèse)
Plasticity of the adrenal stimulus-secretion coupling in arterial hypertension
Increased (elevated and/or prolonged) secretion of catecholamines is a risk factor for pathologies, particularly cardiovascular diseases such as arterial hypertension. Using the genetically hypertensive rat model (SHR), we are studying the plasticity of the determinants of stimulus-secretion coupling, namely the chromaffin cell excitability, the cholinergic synaptic transmission between the splanchnic nerve and chromaffin cells and the gap junctions-mediated electrical coupling between chromaffin cells.
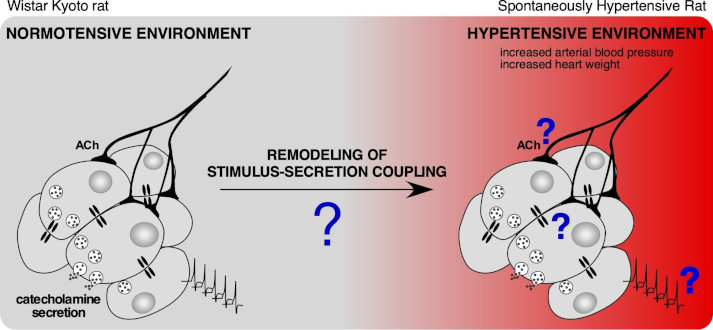
Schematic representation of the stimulation-secretion coupling of the adrenal medullary tissue. What is the impact of a hypertensive environment on the cellular/intercellular mechanisms involved in this coupling? What is the consequence on catecholamine secretion? Adapted from Paillé et al, unpublished results.
Main publications
• Paillé V., et al. (2024) Cell Mol Life Sci, 82, 31.
• Guérineau NC (2024) Vitam Horm, 124, 221-295.
• De Nardi F., et al. (2017) Front Endocrinol, 8, 248.
Funding
• 2014 Angers Loire Métropôle – PI
• 2013 Conseil Général de Maine et Loire – PI
• 2012 Angers Loire Métropôle – PI
• 2010 FRM – PI
• 2010 Région Pays de la Loire – PI
• 2010 Conseil Général de Maine et Loire – PI
• 2010 Angers Loire Métropôle – PI
Collaborations
• Vincent Paillé (Nantes)
• Christian Legros (Angers)
• Daniel Henrion (Angers)
• Jennifer Bourreau (Angers)
• Bertrand Toutain (Angers)
• Joohee Park (Angers)
• Frédéric de Nardi (Angers)
• Dimitri Bréard (Angers)
Alumni
• Vincent Paillé (Post-doctorat)
• Frédéric de Nardi (Post-doctorat)
• Claudie Lefort (Master 2)
Influence of the gut microbiota on the stress response of the adrenal medullary tissue
Chronic stressful situations or dysregulated responses to stress are well-known risk factors to develop neurological pathologies such as depression and anxiety. Via the secretion of stress hormones (catecholamines and glucocorticoids), the adrenal glands hold a key place in the adaptive response to stressors. Elucidating new targets able to affect adrenal secretory function is therefore a current challenge. Growing evidence points the gut microbiota as an emerging target. Reflecting the contribution of the gut microbiota to the stimulus-secretion coupling in chromaffin cells is the finding that the expression of the gene encoding phenylethanolamine N-methyltransferase (PNMT, enzyme converting norepinephrine to epinephrine) is significantly higher in GF stressed rats compared with SPF stressed counterparts.
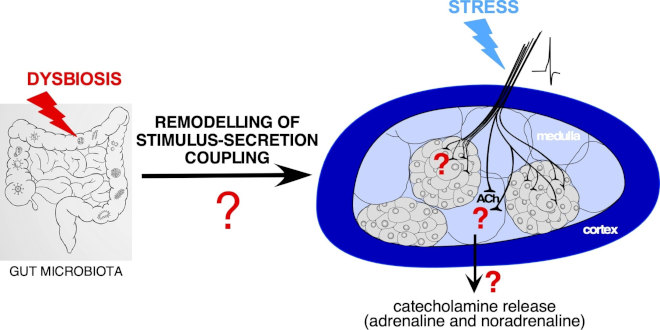
Gut microbiota and stimulus-secretion coupling of adrenal medullary tissue. What is the impact of gut microbiota dysbiosis on the cellular/intercellular mechanisms involved in the stimulus-secretion coupling? What are the consequences for catecholamine secretion and the adaptive response to stress?
Main publications
• Mir HD., et al. (2020) Psychoneuroendocrinology, 119, 104750.
Collaborations
• Sylvie Rabot (Jouy en Josas)
Alumni
• Alexandre Milman (Thèse)


