Team Philippe LORY
Ion channels in neuronal excitability and diseases
Project Deciphering the role of calcium channels in neurodevelopemental diseases
PRINCIPAL INVESTIGATOR

IGF staff involved
Sophie NICOLE
CRHC INSERM
Claire BERNAT
IE CNRS
Arnaud MONTEIL
CRHC CNRS
Amaël DAVAKAN
PhD Student CNRS
Clément MARCHAL
PhD Student UM
Mutations in voltage-gated calcium (Cav) channels are a growing cause of neurodevelopmental diseases. This is the case with spinocerebellar ataxia 42, SCA42ND, caused by de novo, gain-of-function mutations in the CACNA1G gene encoding the Cav3.1 calcium channel, which combines early cerebellar atrophy, microcephaly, epilepsy, with delayed motor and cognitive development. We are studying the electrophysiological and structural consequences of these mutations. Mutations associated with a severe clinical phenotype are gain-of-function and destabilize gating (channel activation and inactivation properties), in particular the recurrent p.A961T mutation, for which a mouse model is being used to study cerebellar motor defects.
We use in vitro (patch-clamp …) and in silico (neuronal modeling, molecular dynamics …) approaches to characterize the consequences of mutations in Cav3 channels, especially mutations located on the intracellular portions of the S5 and S6 segments defining the ‘intracellular gate’. We are developing dedicated animal models for the study of these mutations in order to assess the precise role of these channels in neurodevelopment and related diseases. We are also seeking new and effective therapeutic strategies to treat these complex channelopathies.
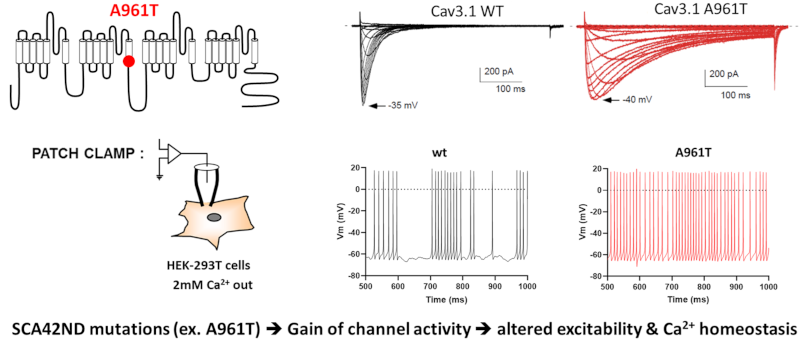
Functional and structural characterization of de novo missense mutations responsible for severe neurodevelopmental diseases, e.g. the p.A961T mutation in Cav3.1 causing SCA42ND. Patch-clamp recordings are used to measure electrophysiological alterations caused by the p.A961T mutation (top right), while neuronal modelling is used to assess the ‘gain-of-function’ nature of the mutation, supported by an increase in the number of action potentials produced by cerebellar neurons (bottom right).
Main publications
• Lory P, et al. (2020) Pflugers Arch. 472(7):831-844 (review).
• Chemin J, Siquier-Pernet K, et al. (2018) Brain. 141(7):1998-2013.
• Dorgans K, Salvi J, et al. (2017) Neurobiol Dis. 106:110-123.
• Cazade M, et al. (2017) Elife. 6:e22331.
• Daniil G, et al. (2016) EBioMedicine 13:225-236.
• Blesneac I, et al. (2015) Proc Natl Acad Sci U S A. 112(44):13705-10.
• Coutelier M, Blesneac I, et al. (2015) Am J Hum Genet. 97(5):726-37.
• Salvi J, et al. (2014) Neurobiol Dis. 68:47-56.
Fundinf
• 2023-2027 – ANR PRC ‘CaCh-Investigation’ project, coordinator (PI)
• 2012-2024- LabEx ICST ‘Ion Channels Science & Therapeutics’, partner
• 2023-2024 – Fondation Connaitre les Syndromes Cérébelleux (CSC), PI
• 2019-2020 – FMR (Fondation Maladies Rares) “animal models” grant, PI
Collaborations
• Vincent Cantagrel (Paris)
• Marion Coolen (Paris)
• Philippe Isope (Strasbourg)
• Xiaojing Cong (Montpellier)
Alumni
• Iulia Blesneac (Postdoc, 2011-2015)


