Team Chris JOPLING
Cardiac Development, Disease and Regeneration
Project Identifying the cellular and molecular mechanisms which drive cardiac regeneration
PRINCIPAL INVESTIGATOR

IGF staff involved
Jourdano MANCILLA ABAROA
PhD Student INSERM
Adèle FAUCHERRE
CRCN CNRS
Thomas MOORE MORRIS
CRCN INSERM
Aurélien DROUARD
IE CNRS
Crystal GUILLEMIN
IE CNRS
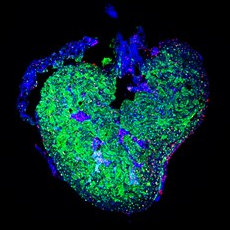
Heart disease has now become one of the most common causes of fatality. Indeed, many European countries report that heart disease is the number one cause of death and disability. Although adult mammals are unable to significantly regenerate their heart, this is not the case for a number of other vertebrate species. Understanding how these animals are capable of regenerating their hearts will provide much needed insights into how this process can be induced in humans in order to reverse the damage caused by myocardial infarction.
Metabolism and cardiac regeneration
Inflammation and cardiac regeneration
Metabolism and cardiac regeneration
Mechanisms driving cardiomyocyte renewal are largely unknown. Among recently identified signaling pathways, the Hippo pathway, myocardial O2 exposure and cardiomyocyte metabolic status are described to dramatically determine cardiomyocyte proliferation abilities and thus are essential for heart regeneration. Nevertheless, how the hypoxic and metabolic status of cardiomyocytes are controlled remains to be determined. Our principal hypothesis is that the interplay between hypoxic and metabolic signaling is of crucial importance for adult heart regeneration.
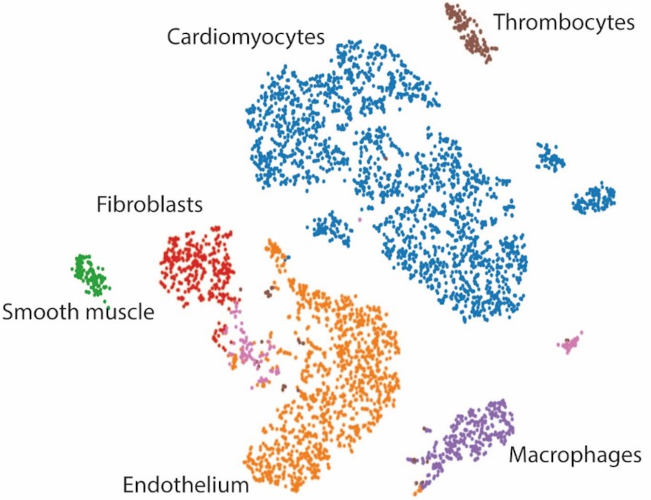
Single nuclei RNA seq analysis of regenerating hearts allows us to identify metabolic genes which regulate cardiomyocyte proliferation.
Main publications
• Jopling C., et al. (2012) Circulation.18;126(25):3017-27.
Funding
• 2022-2025 ANR METABOX – Partenaire
Collaborations
• Francesca Rochais (Marseille)
Inflammation and cardiac regeneration
Macrophages play an important role during cardiac regeneration. Our analysis of the macrophage population revealed that the inflammatory response of resident macrophages followed by the invasion of recruited macrophages appears to be remarkably similar to the response observed in adult mammals after injury, albeit more rapid. Furthermore, we found that recruited macrophages require Mmp14 in order to invade the wound region and promote cardiac regeneration.
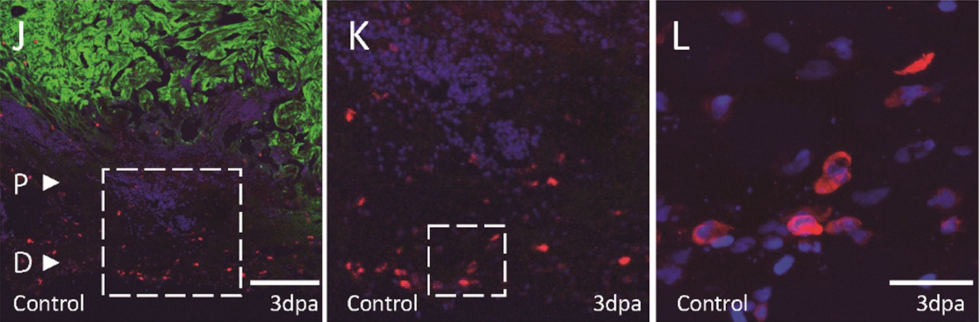
Macrophages (red) invading the wound region of a damaged heart (green) Rolland L., et al. (2023)
Main publications
• Rolland L., et al. (2023) J Mol Cell Biol, 14(10)
Mechanosensation and cardiac regeneration
Myocardial damage caused, for example, by cardiac ischemia leads to ventricular volume overload resulting in increased stretch of the remaining myocardium. In humans, these changes trigger cardiomyocyte hypertrophy resulting heart failure. Conversely, in response to extensive myocardial damage, cardiomyocytes in regenerative species proliferate and completely regenerate the damaged myocardium. Our aim is to determine how changes in mechanical forces due to myocardial damage can be detected by mechanosensors which in turn can trigger cardiac regeneration.
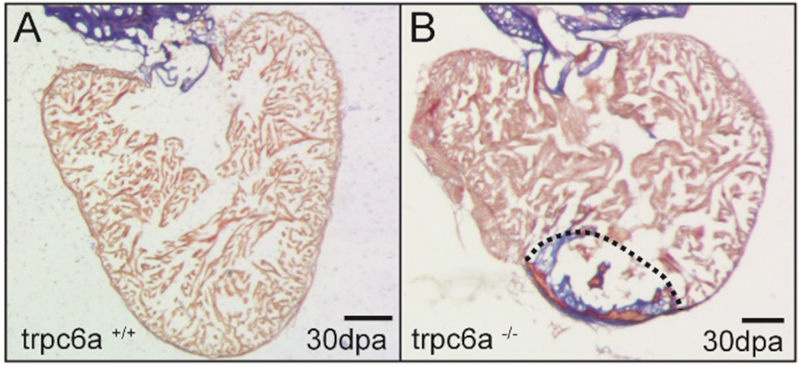
The mechanosensor Trpc6 is required for heart regeneration. The wildtype heart (A) has completely regenerated after 30 days. The Trpc6 KO heart has failed to regenerate and a large scar persists. Rolland L., et al. (2024)
Main publications
• Rolland L., et al. (2024) Front Cardiovasc Med, 10:1186086.
Collaborations
• Francesca Rochais (Marseille)
Cardiotoxicity of environmental pollutants
Recent evidence indicates that environmental pollutants such as Bisphenol A and perfluorinated acids can potentially impact human development and health. Our goal is to determine whether environmental pollutants can cause disrupt cardiac development and physiology. To achieve this, we use in vivo models in conjunction with cutting edge optical mapping and high throughput cardiac and behavioural tests.
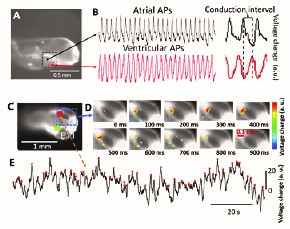
Optical mapping: time series of cardiac and neuronal activity in zebrafish larvae. A) Lateral view of a zebrafish larva. Dotted square indicated the cardiac area. (B): atrial and ventricular action potentials (AP). Inset shows the conduction interval between the atrial and ventricular signals. C,D) Dorsal view of a zebrafish larva. Orange and green cursors indicate typical regions targeted to record brain activity.). E) Example of time series depolarization (targeted region indicated by a dotted line in C,D).
Main publications
• Forner-Piquer I., et al. (2021) Environ Pollut. 22;278:116755.
• Forner-Piquer I., et al. (2021) Chemosphere. 267:128986.
Funding
• 2019 MUSE Pestifish – Partenaire
• 2023 ANSES Optifish – Partenaire
Collaborations
• Nicola Marchi (IGF, Montpellier)
• Angelo Torrente (IGF, Montpellier)


