Team Chris JOPLING
Cardiac Development, Disease and Regeneration
Project Identifying factors involved in cardiac development and disease
PRINCIPAL INVESTIGATOR

IGF staff involved
Victor BERNARD
PhD Student, UM
Chris JOPLING
CRCN, INSERM
Thomas MOORE-MORRIS
CRCN, INSERM
Aurélien DROUARD
IE, CNRS
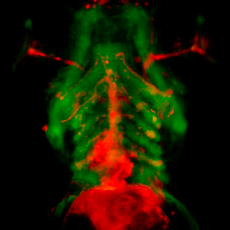
Using a variety of in vitro and in vivo approaches, and in particular the zebrafish model, we investigate the normal and pathological development of the heart. Our aim is to identify factors whose dysregulation leads to congenital heart defects in humans, with a particular focus on the role of hemodynamic forces on extracellular matrix homeostasis and the role of the mechanosensitive Piezo ion channel on cardiac development and rhythmicity.
Mechano-regulation of the cardiac extracellular matrix
Mechano-regulation of the cardiac extracellular matrix
Using a combination of transcriptomic (scRNAseq, snRNAseq), genomic (patient DNA sequencing), in vitro (cell cultures subjected to mechanical forces, electrophysiology) and in vivo approaches in zebrafish and mammals, our aim is to identify the signalling pathways required for the mechanotransduction of hemodynamic forces controlling the expression and homeostasis of the cardiac extracellular matrix and to determine whether they are associated with congenital cardiac defects in humans.
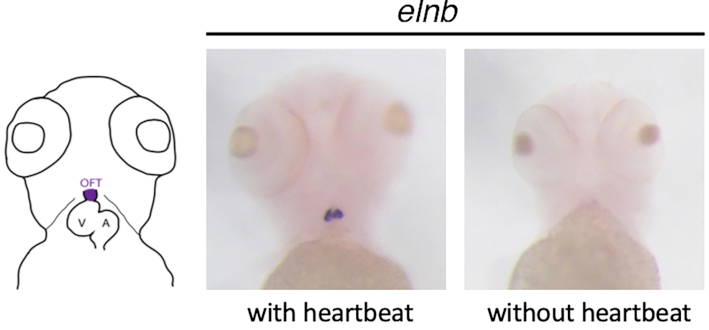
In situ hybridization against elastin b, a component of the extracellular matrix expressed in the cardiac outflow tract. If cardiac contractions (and therefore hemodynamic forces) are inhibited in zebrafish, Elastin is no longer expressed.
Main publications
• Faucherre A., et al. (2020), J Mol Cell Cardiol. 143:51-62.
• Rambeau R. et al. (2017), Int J Cardiol. 15;249:340-343
Funding
• 2012-2025 Labex ICST, ANR-11-LABX-0015 -Member
Collaborations
• Stéphane Zaffran (MMG, Marseille)
Alumni
• Pierre RAMBEAU (PhD)
Mechanosensitive channels and cardiac rhythm
We are interested in the effect of the mechanosensitive Piezo1 ion channel on cardiac rhythmicity. Using several approaches including video microcopy, optical mapping and electrocardiogram recordings, we are studying the effect of activation of this channel on heart rate and its implication in human pathologies such as atrial fibrillation.
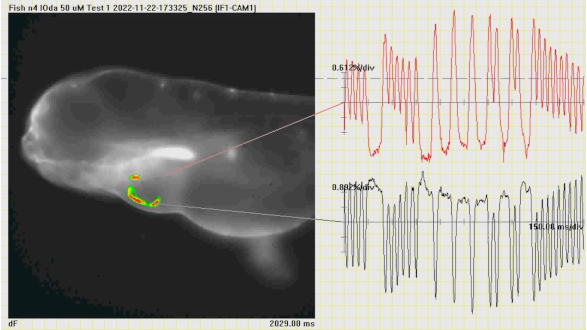
Optical mapping analysis of cardiac action potential diffusion in zebrafishfish treated with a Piezo1 ion channel agonist. From Rolland L., et al. 2033
Main publications
• Rolland L., et al. (2023) Int J Mol Sci. 24(7):6720.
Funding
• 2012-2025 Labex ICST, ANR-11-LABX-0015 – Member
Collaborations
• Philippe Chevalier (Hospices Civils de Lyon)
• Angelo Torrente (IGF, Montpellier)
• Emmanuel Bourinet (IGF, Montpellier)
Alumni
• Laura ROLLAND (PhD)
• Dounia MASKINI (Master)
Genetic variants and congenital heart defects
We are using the zebrafish model to study the pathogenicity of human variants and their involvement in congenital heart disease.
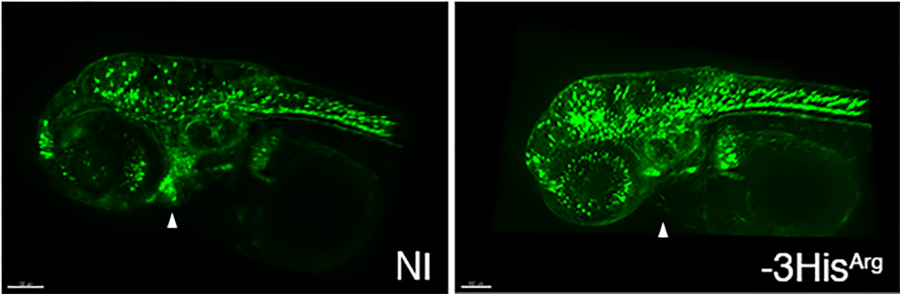
Maximal projection of 3-day-old zebrafish larvae expressing GFP under the control of the sox10 promoter (neural crest). A neural crest cell migration defect is observed in fish injected with the human -3HisArg variant associated with aortic valve defects in humans (NI: not injected). From Odelin G., Faucherre A. et al (2023) Nat Commun. 14(1):1543
Main publications
• Janin A., et al. (2023) Circ Genom Precis Med (3):277-279
• Odelin G., Faucherre A. et al. (2023) Nat Commun. 14(1):1543
• Jaouadi H. et al. (2023) J Transl Med 21(1):160
• Moreau A. (2021) Clin Transl Med. 11(3):e319
• Faucherre A., et al. (2020), J Mol Cell Cardiol. 143:51-62.
Collaborations
• Stéphane Zaffran (MMG, Marseille)
• Philippe Chevalier (Hospices Civils de Lyon)


