Team Emmanuel BOURINET
Calcium channel dynamics and nociception

We study voltage-gated calcium channels, bridging the gap between their molecular function and their impact on the pathophysiology of somatosensation. This functional genomics approach has enabled us to acquire recognized expertise in preclinical research into chronic pain and the sense of touch.
Axis 1
The questions addressed around the role of low-threshold calcium channels concern:
1) their impact on cutaneous and visceral mechanoreceptive sensory neurons
2) their role in spinal neural networks for the integration of sensory information
3) their participation in the excitability of supraspinal circuits activated by mechanoreceptors.
These questions are addressed using preclinical pain models close to the clinic, and using human tissue to validate preclinical work.
Axis 2
In parallel, we are studying autoimmune disorders of peripheral neurons, focusing on demyelinating nodopathies. This approach is directly integrated into the clinic by characterizing sera from patients suffering from these painful pathologies.
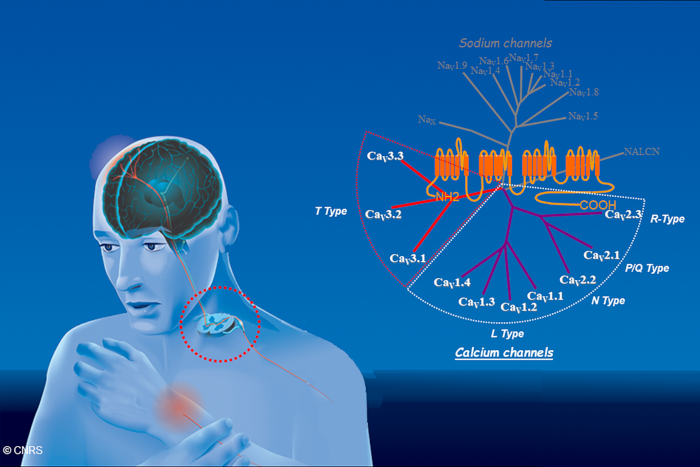
Molecular diversity of calcium channel (CaV) subunits belonging to the 4-domain transmembrane channel super family. The aim of our research is to identify the role of these channels in primary afferent sensory neurons and spinal cord neurons in pain perception.

 IGF Sud 125
IGF Sud 125 04 34 35 92 48
04 34 35 92 48
 IGF Sud 125
IGF Sud 125 04 34 35 92 48
04 34 35 92 48
 IGF Sud 125
IGF Sud 125 04 34 35 92 48
04 34 35 92 48
 IGF Sud 125
IGF Sud 125 04 34 35 92 48
04 34 35 92 48
 IGF Sud 125
IGF Sud 125 04 34 35 92 48
04 34 35 92 48
 IGF Sud 125
IGF Sud 125 04 34 35 92 48
04 34 35 92 48
 IGF Sud 125
IGF Sud 125 04 34 35 92 48
04 34 35 92 48
 IGF Sud 125
IGF Sud 125 04 34 35 92 48
04 34 35 92 48
 IGF Sud 125
IGF Sud 125 04 34 35 92 48
04 34 35 92 48
 IGF Sud 125
IGF Sud 125 04 34 35 92 48
04 34 35 92 48
 IGF Sud 125
IGF Sud 125 04 34 35 92 48
04 34 35 92 48
 IGF Sud 125
IGF Sud 125 04 34 35 92 48
04 34 35 92 48
 IGF Sud 125
IGF Sud 125 04 34 35 92 48
04 34 35 92 48- Yadav, K. J. E. Matson, L. Li, I. Hua, J. Petrescu, K. Kang, M. R. Alkaslasi, D. I. Lee, S. Hasan, A. Galuta, A. Dedek, S. Ameri, J. Parnell, M. M. Alshardan, F. A. Qumqumji, S. M. Alhamad, A. P. Wang, G. Poulen, N. Lonjon, F. Vachiery-Lahaye, P. Gaur, M. A. Nalls, Y. A. Qi, D. Maric, M. E. Ward, M. E. Hildebrand, P. F. Mery, E. Bourinet, L. Bauchet, E. C. Tsai, H. Phatnani, C. E. Le Pichon, V. Menon and A. J. Levine. A cellular taxonomy of the adult human spinal cord. Neuron 2023 111, 328-344 e7. 10.1016/j.neuron.2023.01.007, PMC10044516.
- Huzard, M. Martin, F. Maingret, J. Chemin, F. Jeanneteau, P. F. Mery, P. Fossat, E. Bourinet and A. Francois. The impact of C-tactile low-threshold mechanoreceptors on affective touch and social interactions in mice. Sci Adv 2022 8, eabo7566. 10.1126/sciadv.abo7566, PMC9242590.
- L. Fayad, G. Ourties, B. Le Gac, B. Jouffre, S. Lamoine, A. Fruquiere, S. Laffray, L. Gasmi, B. Cauli, C. Mallet, E. Bourinet, T. Bessaih, R. C. Lambert and N. Leresche. Centrally expressed Cav3.2 T-type calcium channel is critical for the initiation and maintenance of neuropathic pain. Elife 2022 11. 10.7554/eLife.79018, PMC9714965.
- Defaye, M. C. Iftinca, V. M. Gadotti, L. Basso, N. S. Abdullah, M. Cumenal, F. Agosti, A. Hassan, R. Flynn, J. Martin, V. Soubeyre, G. Poulen, N. Lonjon, F. Vachiery-Lahaye, L. Bauchet, P. F. Mery, E. Bourinet, G. W. Zamponi and C. Altier. The neuronal tyrosine kinase receptor ligand ALKAL2 mediates persistent pain. J Clin Invest 2022 132. 10.1172/JCI154317, PMC9197515.
- Beaulieu-Laroche, M. Christin, A. Donoghue, F. Agosti, N. Yousefpour, H. Petitjean, A. Davidova, C. Stanton, U. Khan, C. Dietz, E. Faure, T. Fatima, A. MacPherson, S. Mouchbahani-Constance, D. G. Bisson, L. Haglund, J. A. Ouellet, L. S. Stone, J. Samson, M. J. Smith, K. Ask, A. Ribeiro-da-Silva, R. Blunck, K. Poole, E. Bourinet and R. Sharif-Naeini. TACAN Is an Ion Channel Involved in Sensing Mechanical Pain. Cell 2020 180, 956-967 e17. 10.1016/j.cell.2020.01.033.
- Candelas, A. Reynders, M. Arango-Lievano, C. Neumayer, A. Fruquiere, E. Demes, J. Hamid, C. Lemmers, C. Bernat, A. Monteil, V. Compan, S. Laffray, P. Inquimbert, Y. Le Feuvre, G. W. Zamponi, A. Moqrich, E. Bourinet and P. F. Mery. Cav3.2 T-type calcium channels shape electrical firing in mouse Lamina II neurons. Sci Rep 2019 9, 3112. 10.1038/s41598-019-39703-3, PMC6395820.
- Francois, N. Schuetter, S. Laffray, J. Sanguesa, A. Pizzoccaro, S. Dubel, A. Mantilleri, J. Nargeot, J. Noel, J. N. Wood, A. Moqrich, O. Pongs and E. Bourinet. The Low-Threshold Calcium Channel Cav3.2 Determines Low-Threshold Mechanoreceptor Function. Cell Rep 2015 10, 370-382. 10.1016/j.celrep.2014.12.042.
- Garcia-Caballero, V. M. Gadotti, P. Stemkowski, N. Weiss, I. A. Souza, V. Hodgkinson, C. Bladen, L. Chen, J. Hamid, A. Pizzoccaro, M. Deage, A. Francois, E. Bourinet and G. W. Zamponi. The deubiquitinating enzyme USP5 modulates neuropathic and inflammatory pain by enhancing Cav3.2 channel activity. Neuron 2014 83, 1144-58. 10.1016/j.neuron.2014.07.036.
- Descoeur, V. Pereira, A. Pizzoccaro, A. Francois, B. Ling, V. Maffre, B. Couette, J. Busserolles, C. Courteix, J. Noel, M. Lazdunski, A. Eschalier, N. Authier and E. Bourinet. Oxaliplatin-induced cold hypersensitivity is due to remodelling of ion channel expression in nociceptors. EMBO Mol Med 2011 3, 266-78. 10.1002/emmm.201100134, PMC3377073.
- Bourinet, A. Alloui, A. Monteil, C. Barrere, B. Couette, O. Poirot, A. Pages, J. McRory, T. P. Snutch, A. Eschalier and J. Nargeot. Silencing of the Cav3.2 T-type calcium channel gene in sensory neurons demonstrates its major role in nociception. EMBO J 2005 24, 315-24. 10.1038/sj.emboj.7600515, PMC545807.



