Team Stéphanie BARRÈRE-LEMAIRE – Matteo MANGONI
Cardioprotection, pathophysiology of cardiac rhythm and ischemia
Project Study of the mechanisms of cell death during myocardial ischemia-reperfusion and development of cardioprotective strategies
PRINCIPAL INVESTIGATOR

IGF staff involved
Anne VINCENT
PU UM
Christian BARRERE
IEHC CNRS
Joël NARGEOT
DRCE Emeritus CNRS
Matteo E. MANGONI
DR1 CNRS
Manon MARROT
PhD student CNRS
Mélanie FAURE
PhD student UM
Oscar THABOUILLOT
PhD student, UM
Myocardial infarction (MI) is the leading cause of cardiovascular mortality worldwide. The challenge is to develop cardioprotective strategies to reduce infarct size and the morbidity/mortality rates for patients. Preventing cell death in the acute phase of MI is only possible if the occluded artery is reopened as quickly as possible to restore blood flow. However, myocardial reperfusion is associated with adverse effects due to the abrupt return of oxygen, inducing apoptosis in cardiac cells. Currently, there is no pharmacological treatment to inhibit ischemia-reperfusion (IR) injury and reduce infarct size. Our team is developing cardioprotective approaches to reduce infarct size, reduce the morbi/mortality rates in patients and improve societal costs.
Development of cardioprotective anti-apoptotic peptides
PPARβ⁄δ-mediated preconditioning of mesenchymal stem/stromal cells
Development of cardioprotective anti-apoptotic peptides
We first identified the crucial role of the extrinsic apoptosis pathway downstream of the FAS (First Apoptosis Signal) death receptor in IR injury (Roubille et al, Circulation 2007). Our pharmacological approach uses decoy peptides to inhibit apoptosis by blocking FAS:DAXX or FAS:FADD interaction. When injected at the time of reperfusion, these peptides protect the myocardium over the long term. They are currently evaluated in mice cohorts with co-morbidities (aging, diabetes…). They will be evaluated in regulatory preclinical and clinical trials by the ApoRepair start-up.
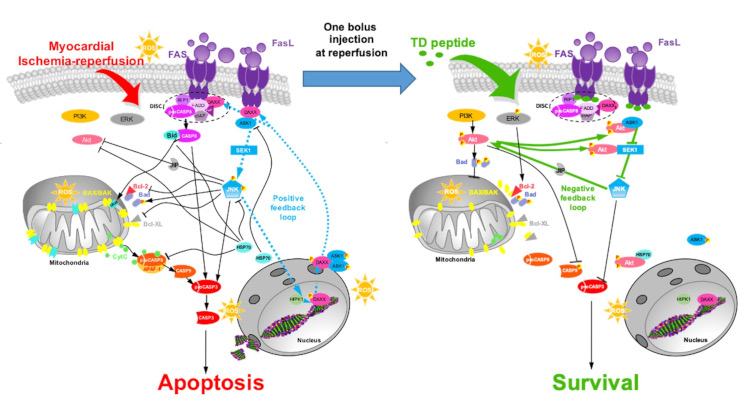
Schematic representation of apoptotic and survival cascades after Tat-DAXXp treatment. (A) Apoptotic signaling cascades activated during myocardial ischemia-reperfusion involving DAXX. (B) Mechanism of action of Tat-DAXXp peptide administered at the onset of reperfusion, leading to inhibition of extrinsic and intrinsic pathways and activation of survival kinases. In both diagrams, feedback loops regulating the nucleo-cytoplasmic ratio of DAXX are shown (from Boisguérin P. et al., Cardiovasc Res 2020).
Main publications
• Fernandez Rico C. et al. (2022). Frontiers in Cardiovascular Medicine; 9:792885.
• Covinhes A. et al. (2020). Scientific Reports;10(1):18116.
• Boisguerin P. et al. (2020). Cardiovascular Research ;116(3):633-644.
• Boisguerin P. et al. (2011). Journal of Control Release;156(2):146-53
• Roubille F. et al. (2007) Circulation; 116 (23):2709-17.
Funding
• 2023-2025 Fédération Française de Cardiologie – Coordinator
• 2021-2024 ANR 2020-NanoCard – Partner
• 2019-2023 Fondation de France – Coordinator
• 2017-2019 Occitanie Region Prematuration Research Contract PepCard – Coordinator
• 2017-2021 ANR 2017-HFADD – Coordinator
• 2014-2017 Prematuration contract SATT AxLR – Coordinator
• 2014-2017 Collaboration research contract CNRS-SERVIER – Coordinator
• 2013-2015 FEDER Contract CardioPep – Coordinatorr
• 2012-2014 ANR 2012-PepFAS – Coordinator
• 2009-2011 ANR 2008-MYOCARDAXX – Coordinator.
Collaborations
• Prisca Boisguérin, PhyMedExp, Montpellier
Alumni
• Carlota Fernandez-Rico (2019-2023)
• Laura Gallot (2012-2013)
• Alicia Franck-Miclo (2009-2012)
• Christelle Redt-Clouet (2009-2011)
• Aurélie Covinhes (2008-2009)
• François Roubille (2005-2008)
Patents
• Licence exclusive d’exploitation de brevet WO 2012/066376.
• Barrere S., Nargeot J., Lebleu B., Boisguérin P., Piot C. EP2982685A1, 2017
• Barrere S., Nargeot J., Lebleu B., Boisguérin P., Piot C. WO 2012/066376, 2012
PPARβ⁄δ-mediated preconditioning of mesenchymal stem/stromal cells
Our cell therapy project targets myocardial ischemia-reperfusion injury with mesenchymal stem/stromal cells (MSC). We have highlighted the crucial role of the PPARβ/δ receptor in the cardioprotective therapeutic properties of MSCs (Nernpermpisooth et al, Front Cardiovasc Med 2021). We showed that MSCs preconditioning with a PPARβ/δ agonist, before transplanting these cells during reperfusion, increased their resistance to transplant stress and their anti-apoptotic properties (Sarre et al, Stem Cell Res Ther 2022).
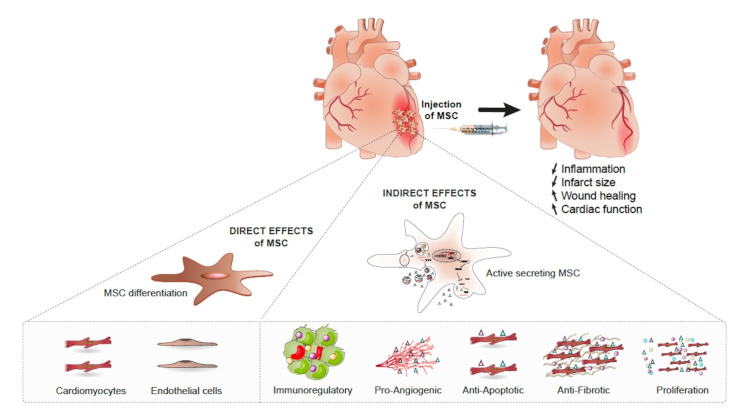
MSC-based therapy of AMI relies on the pleiotropic properties of the cells.
MSC cardioprotective effects can be mediated through direct effects relying on the capacity of MSC to differentiate into cardiac cells or by indirect effects limiting the immune response and apoptosis while increasing angiogenesis and cardiac cell proliferation. When in presence of specific differentiation inducers, MSC differentiate both in vitro and in vivo in cardiomyocytes and endothelial cells which contribute, at least in part, to their ability to repair injured myocardium (from Barrère-Lemaire et al., Physiological Reviews 2024).
Main publications
• Barrère-Lemaire S., et al. (2024). Physiological Reviews;104(2):659-725.
• Sarre C., et al. (2022) Stem cell research & Therapy; 13:167.
• Nernpermpisooth N., et al. (2021) Frontiers Cardiovascular Medicine; 8:681002.
Funding
• 2022-2023 Occitanie Region (Prematuration Research Contract) – PI
• 2021-2024 FRM Postdoctorate contract (for Rafael Contreras)
• 2020-2021 Research collaboration contract – Cell Easy – SATT AxLR- PI
• 2020-2021 SATT AxLR maturation program – PI
• 2017-2020 PhD grant Fonds Elizabeth Brancher (for Charlotte Sarre).
Collaborations
• Farida Djouad, IRMB, Montpellier
• Patricia Luz‑Crawford, Laboratorio de Imunologica Celular y Molecular, Facultad de Medicina, Universidad de los Andes, Santiago, Chile.
• Sarawut Kumphune, Biomedical Engineering Institute, Chiang Mai University, Thailand;
• Nitirut Nernpermpisooth, Department of Cardio-Thoracic Technology, Faculty of Allied Health Sciences, Naresuan University, Phitsanulok, Thailand
• Kantapich Kongpol, School of Allied Health Sciences, Walailak University, Nakhon Si Thammarat, Thailand.
Alumni
• Rafael Contreras (2021-2023)
• Charlotte Sarre (2017-2020)
• Kantapich Kongpol (2019-2020)
• Sanit Jantira (2018-2019)
• Nitirut Nernpermpisooth (2016-2017)
Patents
• Djouad,F., Barrere-Lemaire S., Jorgensen C. WO2022129468A1, 2022.
Searching for new targets for cardioprotection
Our team (part of the ICST LabEx) aims to develop therapeutics targeting intracellular calcium signalling, which plays a key role in survival, contraction, pacemaking and cell death mechanisms. We investigated the endogenous mechanisms mediating ischemic posconditioning. We have identified Cav1.3 (Delgado et al., Front Cardiovasc Med 2023), mGLUR1 (Vincent et al., Cardiovasc Res 2017) as targets of cardioprotection and developed a multi-target approach based on MLC901 TCM (Vincent et al., Sci Rep 2017).
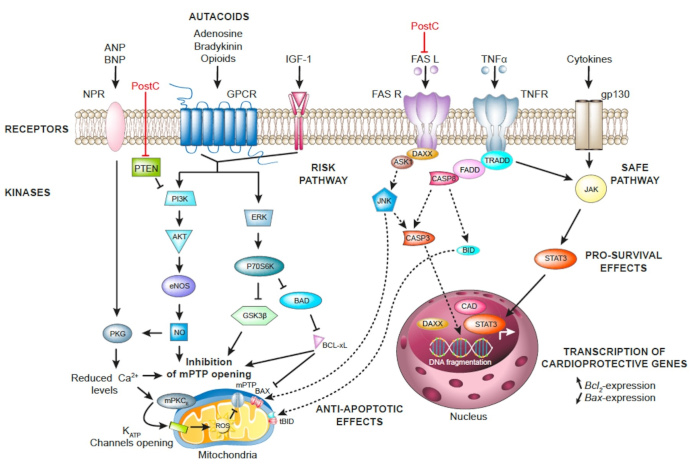
Endogenous mechanisms of cardioprotection.
Upon postconditioning (PostC) stimulus, autacoids (adenosine, bradykinin, endogenous opioids) are released and bind to their specific receptors belonging to the superfamily of GPCR. RISK signaling pathways comprising survival kinases such as PI3K/AKT and ERK1/2 (p42/44) are downstream cascades activated upon PostC stimulus. PTEN is a powerful negative regulator of PI3K activity. The final endpoint of this intracellular cascade is the inhibition of MPTP opening via inhibition of GSK3β, increased NO (via AKT) and PKG activity (via natriuretic peptides receptors) and reduced intracellular Ca2+ levels. The opening of mitochondrial KATP channels is part of the endpoints of the posconditioning mechanisms.
The SAFE pathway mediated by JAK-STAT3 is also responsible for postconditioning induced-cardioprotective effects. STAT3 relocates to the nucleus in order to control the transcription of cardioprotective genes such as Bcl-XL. The cardioprotective effects of PostC results also from the inhibition of apoptotic pathways (dotted lines) mediated by the extrinsic pathway involving death receptors such as FAS and TNF receptors.
Main publications
• Mesirca P. et al. (2024). Nature communications; 15(1):54.
• Kumphune S. et al. (2023). Frontiers in cardiovascular medicine; 10:1260800
• Delgado-Betancourt V. et al. (2023). Frontiers in cardiovascular medicine;10:1134503
• Vincent A. et al. (2017). Scientific Reports;7(1):14701.
• Vincent A. et al. (2017). Cardiovasc Research ;113(6):644-655.
• Vincent A. et al. (2012). Cardiovasc Research; 94(2):351-8.
Funding
• 2020-2025 ANR 2011-LABEX ICST
• 2020-2021 PHC SIAM Campus France.
• 2015-2016 Contrat collaboration MOLEAC EUROPE.
• 2014-2015 PHC SIAM Campus France.
• 2011-2020 ANR 2011-LABEX ICST
Collaborations
• Catherine Heurteaux, IPMC, Sophia Antipolis
• Michel Lazdunski, IPMC, Sophia Antipolis
• Sarawut Kumphune, Biomedical Engineering Institute, Chiang Mai University, Thailand
Alumni
• Kroekkiat Chinda (2016-2017)
• Delgado-Betancourt (2012-2015)


