Team Stéphanie BARRÈRE-LEMAIRE – Matteo MANGONI
Cardioprotection, pathophysiology of cardiac rhythm and ischemia
Project Modulation of cardiac activity by the autonomic nervous system: effect of ageing and application to the ethology of marine species
PRINCIPAL INVESTIGATOR

IGF staff involved
Manon MARROT
PhD student CNRS
Mélanie FAURE
PhD student UM
Jeremias CHOUSIDIS
Postdoc CNRS
Pierre FONTANAUD
IECN CNRS
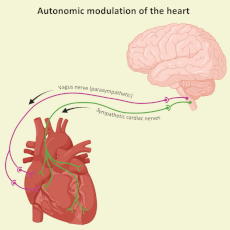
The autonomic nervous system (ANS) directs the body’s involuntary functions and modulates heart rate according to the body’s needs. This modulation, mediated by the sympathetic and parasympathetic branches of the ANS, underlies variability in heart rate that help control metabolism, respond to stress or reduce cardiac activity at rest. Therefore, heart rate variability (HRV) provides essential information on the functioning of the cardiovascular system, heart-brain interaction and the perception of external stimuli. We use HRV to study how aging affects cardiac activity and to investigate sensory perception in marine species, such as cetaceans, turtles and seabirds.
Effect of aging on cardiac pacemaker activity and its autonomic modulation
Application of heart rate variability to the behavioral study of cetaceans and other marine species
Effect of aging on cardiac pacemaker activity and its autonomic modulation
Ageing is a key factor in cardiovascular diseases. It causes a gradual reduction in cardiac functions that can impair the body’s aerobic and cognitive capacities. To date, researchers have focused on abnormalities in the intrinsic activity of the heart (pacemaker and contractile) during ageing. We are interested in the effects of ageing on the cardiac autonomic modulation. To do this, we are studying the small primate lemur Microcebus murinus to improve translatability of basic science results to human pathologies related to aging.

Phenotypic comparison between young (left) and old (right) Microcebus Murinus.
Main publications
• DiFrancesco ML, et al. (2023) Sci Rep. 13(1):3054. doi: 10.1038/s41598-023-29723-5
Funding
• 2020-2025 Fondation Leducq TNE 19CV03 FANTASY
• 2024-2025 Soutien à la Recherche University of Montpellier
Collaborations
• Nadine Mestre-Francès (Montpellier)
• Halina Dobrzynski (Manchester)
• Pierre Sicard (Montpellier)
Alumni
• Yasmine Colombani (Master 2, 2023)
Application of heart rate variability to the behavioral study of cetaceans and other marine species
Marine animals such as cetaceans, turtles and penguins have developed adaptations to intermittent breathing that make their cardiac physiology extreme and fascinating. Heart rate variability (HRV), determined by unconscious responses to stress and other stimuli, is a good indicator of how these animals perceive the surrounding world. Based on this principle, we are using HRV to better understand the behavior of different marine species and to obtain comparative physiological information that will help us to better understand human physiology.
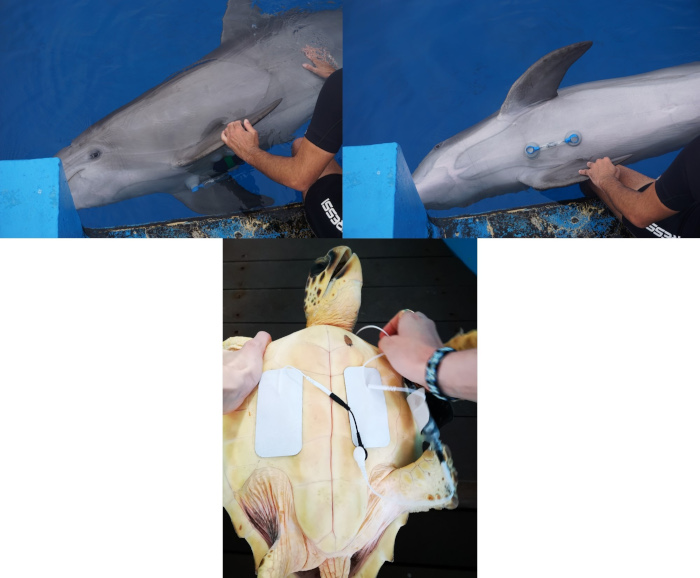
Electrocardiogram recordings on a common dolphin (Tursiops truncatus) and a loggerhead sea turtle (Caretta caretta)
Main publications
• Fahlman A., et al. (2023) Front Physiol 21:14:1234432. doi: 10.3389/fphys.2023.1234432.
• Bonadonna F., Caro S.P., Belle S., Torrente A.G. 2024 Blue petrel electrocardiograms measured through a dummy egg reveal a slow heart rate during egg incubation. Animal Biotelemetry, in press
Funding
• 2023-2026 ANR DicWoc
• 2023 Explore 4 University of Montpellier
Collaborations
• Aurelie Celerier (Montpellier)
• Francesco Bonadonna (Montpellier)
• Andreas Fahlman (Valencia)
Alumni
• Marien Perry (Master 2, 2023)
Study of cardiac function and of the electrical activity in excitable tissues using optical voltage mapping
As a complement to cellular electrophysiology, optical mapping enables to visualize in 2 dimensions the activity of cellular networks and study the generation and conduction of electrical impulses (voltage changes) and the release of intracellular calcium in excitable tissues such as the heart, brain and muscles. This technique, with its high temporal resolution, has enabled us to study ex vivo the physiology of the whole heart of mice, Microcebus murinus and zebrafish to gain a better understanding of the mechanism of cardiac pacemaker activity.

Top panel: Optical voltage mapping system. Bottom left panel: voltage isochrone showing in red the location of the generation of the electrical impulse and its conduction thanks to the different color degrees. Bottom right panel: Optical action potentials recorded in the area of the sinus node (SAN) and the right (RA) and left (LA) atria.
Main publications
• Micou S. et al. (2023) Environmental Technology & Innovation 31,103196.
• Rolland L. et al Int J Mol Sci. (2023) 4;24(7):6720.
• Fossier L. et al (2022) J Am Coll Cardiol. 6;80(23):2205- 2219
• Baudot M, et al. Sci Rep. (2020);10(1):18906
• Torrente AG, et al. (2015) PNAS 201505670.
Funding
• 2023-2025 OptoFish Anses (PNR EST)
• Espoir en Tête FRC 2022
• 2021 – 2022 Soutien à la recherche University of Montpellier
Collaborations
• Nicola Marchi (Montpellier)
• Adele Faucherre (Montpellier)
• Chris Jopling (Montpellier)
• Juan Francisco Llopis Borras (Albacete)
Alumni
• Matthias Baudot (PhD, 2016-2019)
• Lucille Fossier (Master, 2018)


