Team Stéphanie BARRÈRE-LEMAIRE – Matteo MANGONI
Cardioprotection, pathophysiology of cardiac rhythm and ischemia
Project Ion channels in the genesis and regulation of heart rate and rhythm
PRINCIPAL INVESTIGATOR

IGF staff involved
Isabelle BIDAUD
IECE Inserm
Pietro MESIRCA
CRCN Inserm
Eleonora TORRE
Postdoc CNRS
Walma VASCONCELOS
Postdoc CNRS
Mélanie FAURE
PhD student UM
Manon MARROT
PhD student CNRS
Nadia MEKRANE
PhD student UM
Eliane MORAIS-PINTO
PhD student UM
Andrea CARA
AI CNRS
Leïla TALSSI
IE Inserm
Pinidmontree CHIRUTCHAYA
PhD student (Univ.Chulalongkorn, Thaïland)
Pierre FONTANAUD
IECN CNRS
Angelo TORRENTE
CRCN CNRS
Anne Vincent
Professor UM
Joël NARGEOT
DR Emeritus CNRS
Christian BARRERE
IECE CNRS
Stéphanie BARRERE-LEMAIRE
DR2 CNRS
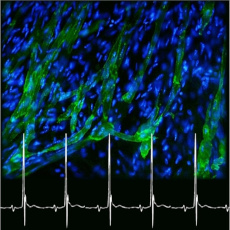
We develop a long-term research project to unravel the specific role of ion channels and their regulatory pathways in the genesis and regulation of heart automaticity and pacemaker activity during lifetime and ontogenesis. The project involves four main axis: (1) the biophysical, pharmacological et structural study of the properties of ion channels involved in automaticity of pacemaker myocytes (green cells in the picture); (2) the study of the functional role of ion channels using a genetic-based approach in mice; (3) the development of models of primary (inherited) and secondary (acquired) dysfunctions of heart automaticity and heart rhythm, and (4) the study of cellular models of heart automaticity based on differentiation of adult or induced stem cells.
Ion channels and regulatory signalling pathways in cardiac pacemaker myocytes
Calcium channels in cardiac arrhythmias
Differentiation of pacemaker myocytes derived from adult skeletal muscle derived stem cells
Ion channels and regulatory signalling pathways in cardiac pacemaker myocytes
We employ a worldwide unique collection of genetic models of heart automaticity in which one or more ion channels involved in pacemaker activity have been ablated or modified. We develop multi-scale approach including cellular electrophysiology, electrophysiology and imaging in intact hearts and pacemaker tissue, as well as in vivo telemetric electrocardiogram. We integrate our results in computation models of heart automaticity and cellular pacemaker activity.
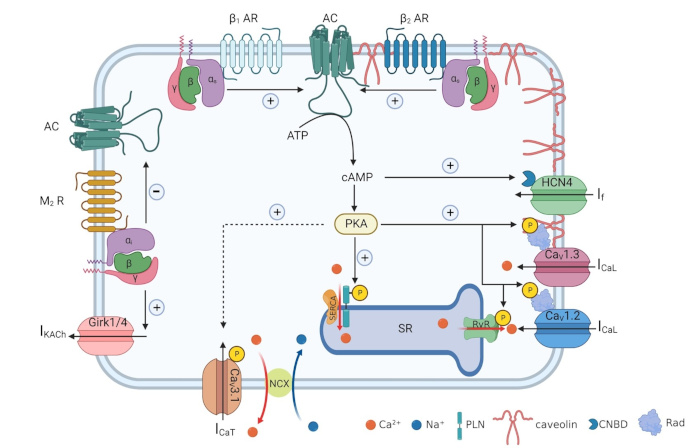
Schematic diagram of adult pacemaker myocyte displaying the main ion channels involved in pacemaker activity and regulatory pathways involved in the regulation of pacemaking by autonomic nervous system.
Main publications
• Mesirca et al. (2024) Nat Commun, 15:54. doi: 10.1038/s41467-023-43502-w.
• Louradour et al. (2022) Cells, 25,11,1114. doi: 10.3390/cells11071114.
• Baudot et al. (2020) Sci Rep, 10, 18906. doi: 10.1038/s41598-020-76049-7.
• Toyoda et al. (2017) Sci Rep, 7, 7869. doi: 10.1038/s41598-017-08191-8.
• Torrente et al. (2016) Cardiovasc Res, 109, 451, doi: 10.1093/cvr/cvw006.
Funding
• 2024-2027 ANR “L-type Cav1.3 (α1D) calcium channels as targets for controlling heart failure related ventricular arrhythmias” (CANTATA), Partenaire
• 2024-2027 Equipe Fondation pour la Recherche Médicale (FRM).
• 2024-2027 ERANet – ERA4Health-CARDINNOV – Partenaire.
• ANR 2023-2027. Defining the ionic bases of the fight-or-flight response of heart rate (IFOR) – Coordonateur
• 2023-2025 France-Canada Research Fund. Establishing a new model for understanding mechanisms driving cardiac functional performance. co-PI avec Emily Standen, Univ. Ottawa.
• 2020-2025 Fondation Leducq Transatlantic Network of Excellence “Fighting Against Sinus Node Dysfunction and Associated Arrhythmia”s (FANTASY) – Coordonateur Européen.
• Labex Ion channels Science and Therapeutics (ICST).
Collaborations
• Alicia D’Souza (Imperial College London, U.K.)
• Mark R. Boyett (Univ. Bradford, U.K.)
• Anna Moroni et Dario DiFrancesco (Univ. Milan, Italie)
• Steven O. Marx (Columbia Univ., NY, USA)
• Ana-Maria Gomez (Univ. Paris-Saclay, Orsay, France)
• Michel De Waard (l’Institut du Thorax, Nantes)
• Peter J Mohler et Vadim Fedorov (Univ. of Ohio, Columbus, USA)
• Dirk Isbrandt (Univ. Cologne, Allemagne)
• Gregoire Vandecasteele (Univ. Paris-Saclay, Orsay, France)
• Marta Campiglio and Valentina Di Biase (Medical University Innsbruck, Austria)
• Amy Lee (Univ. du Texas)
• Bernard Attali (Univ. de Tel Aviv, Israel)
• Emily Standen (Univ. d’Ottawa, Canada)
Alumni
• Julien Louradour (PhD, 2018-2021)
• Matthias Baudot (PhD, 2015-2018)
Calcium channels in cardiac arrhythmias
Ion channels such as hyperpolarisation-activated HCN4 or L-type Cav1.3 underlie heart automaticity and cardiac conduction under physiological conditions. While their functional expression is generally absent or reduced in the adult healthy myocardium, specific genetic or physiological conditions such as training, circadian rhythms, ageing, or pathologies such as atrial fibrillation and heart failure can alter ion channels’ expression pattern and function, leading to arrhythmias. Our goal is to understand these mechanisms of altered ion channels expression and regulation to target arrhythmias.
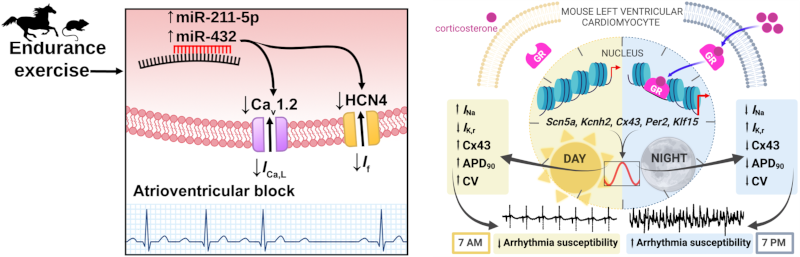
Image representing a physiological condition, chronic exercise training (left), or circadian rhythm (left) regulate the expression of ion channels. As a result, exercise training is responsible for an increased risk of bradycardia or atrio-ventricular block by activating the expression of micro-RNAs that reduce channel expression. The circadian activity of the glucocorticoid receptor (GR) is responsible for the epigenetic regulation of cardiac sodium channels and connexins, which favours the generation of ventricular arrhythmias on awakening.
Physiological conditions such as exercise training (left) and circadian rhythms (right) regulate ion channels expression. Chronic exercise training underlies increased risk of bradycardia and atrioventricular block in athletes, by activating transcription of micro-RNAs downregulating ion channels expression. Circadian activity of glucocorticoid receptors (GR) underlies diurnal epigenetic regulation of expression of cardiac sodium channels and connexins, favoring onset of ventricular arrhythmias in the morning time.
Main publications
• Tikomirov et al. (2024) Circ Res, 134:1306-1326, doi: 10.1161/CIRCRESAHA.123.323464
• Al-Othman S, et al. (2024) Heart Rhythm. S1547-5271(24)00222-4. doi: 10.1016/j.hrthm.2024.02.050.
• Shrestha N, et al. (2023) Proc Natl Acad Sci U S A. e2210152120. doi: 10.1073/pnas.2210152120. Epub 2023 Jul 5.
• Mesirca et al. (2021) Annu Rev Pharmacol Toxicol, 61:757-778. doi: 10.1146/annurev-pharmtox-031120-115815
• Mesirca et al. (2016) PNAS,113, E932-41. doi: 10.1073/pnas.1517181113.
Funding
• 2024-2027 ERA-NET: ERA4HEALTH. SK4 K+ channel blockers: targeting calmodulin-PIP2 interface as new mechanism-based treatment of atrial fibrillation and heart failure – Partner
• 2024-2027 Equipe Fondation pour la Recherche Médicale
• 2020-2025 Fondation Leducq TNE Fighting Against Sinus Node Dysfunction and Associated Arrhythmias (FANTASY) – European Coordinator.
Collaborations
• Alicia D’Souza (Imperial College London, U.K.)
• Mark R. Boyett (Univ. Bradford, U.K.)
• Henggui Zhang (Univ. Manchester, U.K.)
• Francis Marchlinski/Matthew Hyman (Univ. of Pensylvania, USA)
• Eric Schulze-Bahr (Univ. Münster, Allemagne)
• Peter J Mohler et Vadim Fedorov (Univ. of Ohio, Columbus, USA)
• Jean-Pierre Benitah (Univ. Paris Saclay, Orsay, France)
• Bernard Attali (Univ. de Tel Aviv, Israel)
Alumni
• Matthias Baudot (PhD, 2015-2018)
• Antony Chung You Chong (PhD, 2016-2019)
Differentiation of pacemaker myocytes derived from adult skeletal muscle derived stem cells
We identified a new population of adult stem cells derived from the skeletal muscle (MDSCs) showing multi-lineage differentiation including spontaneous differentiation of myocytes endowed of pacemaker activity and sharing molecular markers with native cardiac pacemaker myocytes. We study the pacemaker mechanism of these myocytes at differentiated stage and their capacity to improve heart rate and chemotropic response in models of bradycardia and autonomic incompetence.
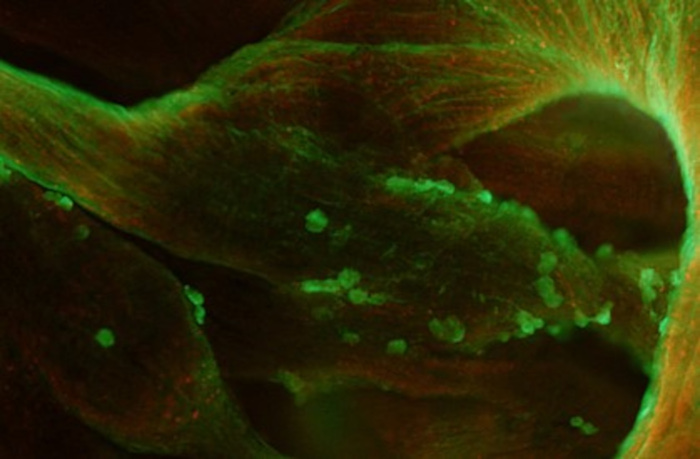
Close-up view of right atrium subregion showing fluorescently labelled MDSCs (green cells) after intravenous administration. MDSCs spontaneous home in the sinoatrial node and atrial trabeculations. In the example here MDSCs are still in the proliferative stage (note cells in mitosis phase of cell cycle). Picture: Anne Fernandez (IGH).
Main publications
• Mesirca et al. (2024) Biox XIV, https://doi.org/10.1101/393512
Funding
• 2023-2027 ANR. Defining the ionic bases of the fight-or-flight response of heart rate (IFOR) – Coordonateur
• 2020-2025 Fondation Leducq TNE Fighting Against Sinus Node Dysfunction and Associated Arrhythmias (FANTASY) – European Coordinator
Collaborations
• Anne Fernandez (IGH, Montpellier)
• Ned Lamb (IGH, Montpellier)
• Daria Mamava (INM, Montpellier)
• Nicola Arsic (IGMM, Montpellier)
• Alicia D’Souza (Imperial College London, U.K.)
Alumni
• Matthias Baudot (PhD, 2015-2018)
• Mattia Lorenzo Di Francesco (Postdoc, 2012-2016)


