SELECTIVE BLOCKADE OF CaV1.2 VERSUS CaV1.3 L-TYPE Ca2+ CHANNELS BY THE BLACK MAMBA TOXIN CALCISEPTINE
L-type voltage-gated calcium channels are involved in multiple physiological functions. The heart expresses two L-type Ca2+ channels isoforms, Cav1.2 and Cav1.3. Cav1.2 is ubiquitously expressed in the heart ventricular and supraventricular chambers, as well as in the sinoatrial (SAN) and atrioventricular (AVN) nodes. Cav1.2 is the predominant L-type isoform in adult ventricle, where it couples excitation to contraction and constitutes the target of the cAMP/PKA mediated positive inotropic effect on cardiac contractility exerted by catecholamines. Cav1.3 is expressed in the atria and is strongly expressed in the SAN and AVN. Cav1.3 plays a major role in SAN pacemaker activity and impulse conduction through the AVN. Despite the importance of dissecting the physiopathological roles of Cav1.2 and Cav1.3 channels in cardiac myocytes and neuronal cells, no selective pharmacological tool is currently available. Animal venoms constitute an immense reservoir for identification of new toxins and toxin-derived tools for identifying the physiological function of ion channels in various tissues.
A collaborative study involving three IGF teams (team “Cardioprotection, pathophysiology of heart rhythm and ischemia” led by Stéphanie Barrère and Matteo Mangoni, team “Calcium channel dynamics and nociception” led by Emmanuel Bourinet and team “Ion channels in neuronal excitability and diseases” led by Philippe Lory), the Institute of Molecular and Cellular Pharmacology (Valbonne, Université Côte d’Azur) and the Department of Physiology at the National University of Singapore, has shown that Calciseptine (Cas), a polypeptide from the venom of the black mamba, selectively blocks Cav1.2 channels, without affecting Cav1.3 channels. Cas potently decreases cardiac contractility without affecting heart rate of Langendorff perfused mouse heart. While Cas totally blocks ICaL (Cav1.2) in ventricular myocytes, it partially inhibits ICaL current (Cav1.2+Cav1.3) in SAN myocytes from wildtype mice and fully blocks ICaL in myocytes from Cav1.3−/− counterparts, where only Cav1.2 is functional. The selectivity of Cas blockade of Cav1.2 versus other calcium channels has been confirmed in HEK-293T expressing recombinant Cav1.2, Cav1.3 (both Cav1.342a and Cav1.342 splice variants), T-type Cav3.1, N-type Cav2.2 and P/Q-type Cav2.1 channels.
In conclusion, this study shows that Cas selectively blocks Cav1.2 channels in the heart, providing an important tool in the panel of calcium channel modulators to help dissect the role of this channel isoform in heart physiology and automaticity but also in neuronal cells in which each isoform is mediating specific physiological functions.
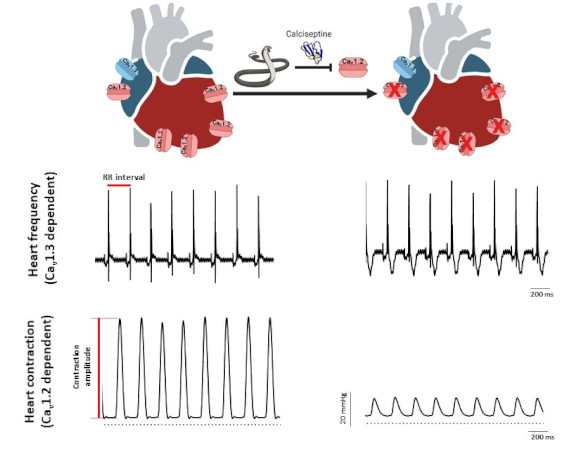
Heart rate and ventricular contraction in Cas perfused heart. Representative ECG traces (top) and pressure recordings (bottom) from isolated perfused heart in control condition (left) and in the presence of calciseptine (right).


