The nuclear receptor REV-ERBα is implicated in the alteration of β-cell autophagy and survival under diabetogenic conditions
Pancreatic β-cell failure in type 2 diabetes mellitus (T2DM) is associated with impaired regulation of autophagy which controls β-cell development, function, and survival through clearance of misfolded proteins and damaged organelles. However, the mechanisms responsible for defective autophagy in T2DM β-cells remain unknown.
Since recent studies identified circadian clock transcriptional repressor REV-ERBα as a novel regulator of autophagy in cancer, this study conducted by Dr. Safia Costes (Dalle-Renard Team, Institute of Functional Genomics, Montpellier) in collaboration with Pr. Aleksey Matveyenko (Mayo Clinic, Rochester, MN, USA) , set out to test whether REV-ERBα-mediated inhibition of autophagy contributes to the β-cell failure in T2DM. This study provides evidence that common diabetogenic stressors (e.g. glucotoxicity and cytokine-mediated inflammation) augment β-cell REV-ERBα expression and impair β-cell autophagy and survival. Notably, pharmacological activation of REV-ERBα was shown to phenocopy effects of diabetogenic stressors on the β-cell through inhibition of autophagic flux, survival, and insulin secretion. In contrast, negative modulation of REV-ERBα was shown to provide partial protection from inflammation and glucotoxicity-induced β-cell failure.
Finally, using bioinformatic approaches, Brown et al. provide further supporting evidence for augmented REV-ERBα activity in T2DM human islets associated with impaired transcriptional regulation of autophagy and protein degradation pathways.
In conclusion, this study reveals a previously unexplored causative relationship between REV-ERBα expression, inhibition of autophagy and β-cell failure in T2DM.
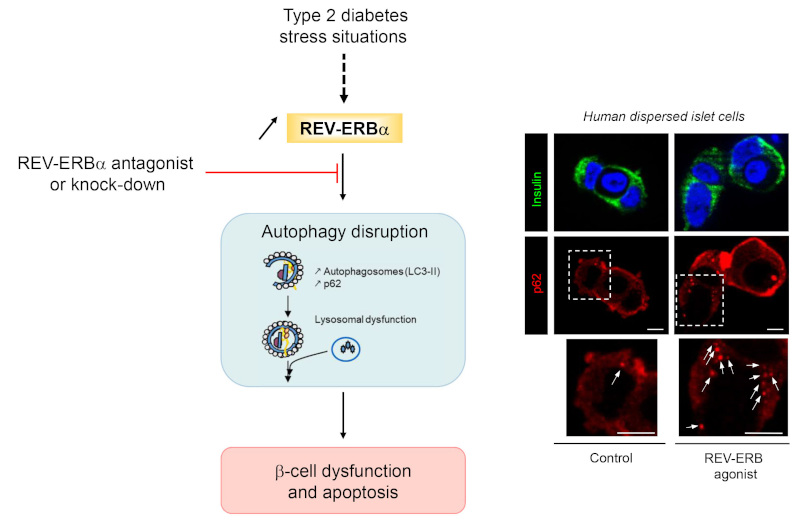
REV-ERBα in pancreatic β-cells.
On the right, p62-positive cytoplasmic inclusions (marker of autophagy disruption) are increased in human β-cells exposed to REV-ERBα agonist. p62 protein levels were assessed by immunofluorescence (p62, red; insulin, green; nuclei, blue) in human dispersed islet cells.


