Glutamate is the most abundant transmitter at the excitatory synapses and acts on receptor-channels within milliseconds. Glutamate also acts on slower-acting receptors that regulate many synapses, and represent targets for new drugs. Eight of such receptors are already known, consisting of two identical entities. Our group just report that there are also glutamate receptors made of two different entities, revealing the existence of 16 new receptors with specific properties. These results are presented in the journal Nature Chemical Biology.
Glutamate is known for its gustatory action at the origin of the “umami” flavor. This amino acid is also the main neurotransmitter in our brain used by more than three quarters of the synapses. Glutamate activates ion channels responsible for rapid excitatory transmission. For example, it allows the extremely rapid analysis of visual information, which will lead to an essential movement in two tenths of a second. These excitatory properties can, in case of excess, lead to epileptic seizures or even neuron toxicity. Fortunately, there are processes that can be used to limit excess activity of the excitatory synapses. One of the most important is the action of glutamate on regulatory receptors, called metabotropic or mGlu receptors, which transmit their signaling into the cell via metabolic pathways. These mGlu receptors measure ambient glutamate and regulate the activity of many synapses. These receptors were discovered almost 40 years ago at the IGF, and the 1990s revealed eight genes encoding these receptors. These receptors are composed of two subunits that move relative to each other when glutamate binds to them, leading to the transmission of information within the cell. Since this discovery, they were considered to be made up of two identical subunits.
But in 2011, our group demonstrated that mGlu receptors could be composed of two different subunits, with specific compositions, and particular functional properties. These data suggested the existence of 16 additional heterodimeric mGlu receptors, one of whose structures was resolved. Finally, functional evidence of their existence at certain synapses was provided. But their existence in the brain remained to be demonstrated.
In collaboration with the chinese team of Prof. J. Liu (Huazhong University, Wuhan, China), our group has demonstrated the presence of the mGlu2-4 heterodimer in different brain regions in mice. They have developed an approach based on the measurement of a light signal linked to the proximity between two innovative antibodies called nanobodies, one directed against mGlu2 and the other against mGlu4. Developed from llamas, these nanobodies are 10 times smaller than a conventional antibody. This approach allowed the quantification of mGlu2 and mGlu4 subunits as homodimers and heterodimers in the brain. These results show that there are more heterodimeric mGlu2-4 receptors than mGlu4-4 receptors in the brain, except in the cerebellum.
These data unequivocally demonstrate the existence of mGlu receptors consisting of two different subunits, reinforcing the idea that there are more mGlu receptor subtypes than previously expected. This opens up the possibility of more specific targeting of certain synapses and more precise therapeutic actions. These heterodimeric mGlu receptors therefore represent new potential targets. They open new avenues of research for neurological and psychiatric diseases in which mGlu receptors play an important role. The way is open for the search for new molecules that can act specifically on these new mGlu receptors.
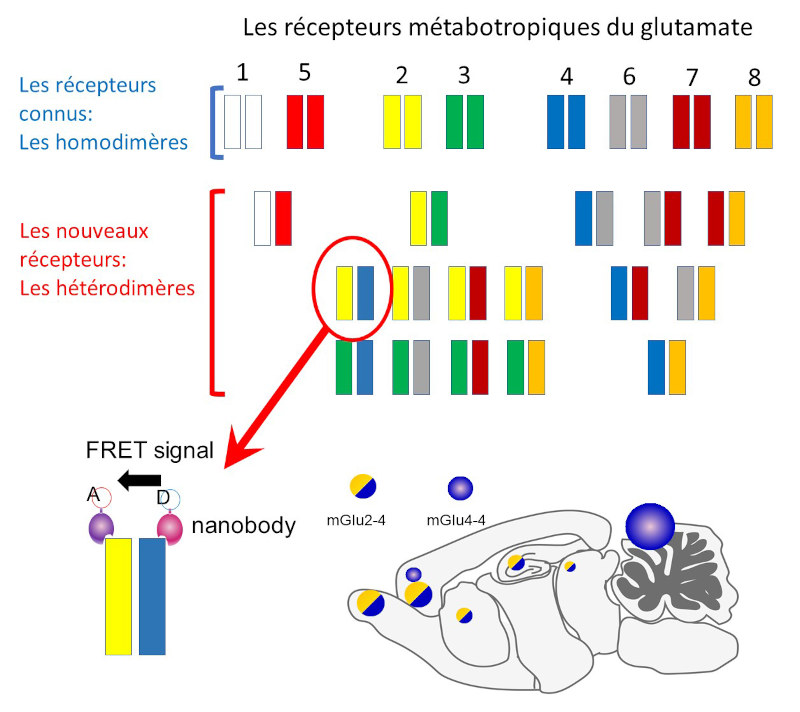
New metabotropic glutamate receptors consisting of two different entities (heterodimers), mGlu2 and mGlu4, have been discovered in the mouse brain.
This discovery suggests that other heterodimeric mGlu receptors exist in the brain in addition to the 8 homodimeric (1-8) mGlu receptors already known (a). The mGlu2-4 heterodimers were detected by measuring a light signal related to the proximity between two innovative antibodies called nanobodies, one directed against mGlu2 and the other against mGlu4 (b). The study shows that there are more heterodimeric mGlu2-4 receptors than mGlu4-4 receptors in the brain, except in the cerebellum (c).


