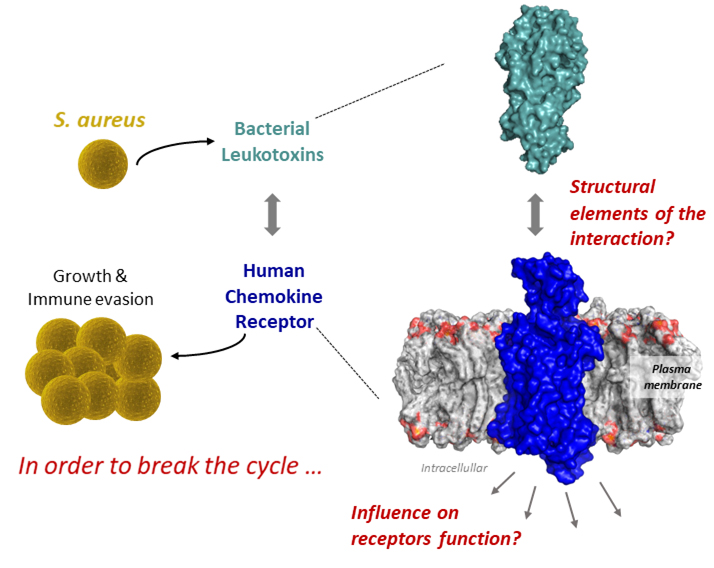
The rise of multidrug resistant strains of Staphylococcus aureus (SA) represents a major threat to public health. To combat resistant strains, we urgently need to target novel molecular mechanisms essential for SA growth and pathogenesis. One such essential mechanism is bacterial leukotoxins secreted by SA that binds to host receptors, mainly host chemokine receptors present at the surface of myeloid and erythroid cells, leading to
In order to understand how these toxins influence their targeted receptor, the team of Sébastien Granier at the IGF (CNRS/INSERM/University of Montpellier), in a study co-directed by Chérine Bechara, developed an innovative integrative approach of pharmacology and structural mass spectrometry. This work published in Proc. Natl. Acad. Sci. describes the molecular mechanisms of toxin/receptor binding and the effects on receptor’s conformation, revealing therefore how the toxins modifies the structure of the receptor to hijack its activity.
This study constitutes the first step towards the development of novel inhibitors to tackle SA growth and virulence by blocking toxins binding to host receptors.
Structure and function of leukotoxin/receptor complexes: a new therapeutic window to inhibit SA infection


