GLP-1 AND GIP RECEPTORS SIGNAL THROUGH DISTINCT Β-ARRESTIN 2-DEPENDENT PATHWAYS TO REGULATE PANCREATIC Β CELL FUNCTION
GLP-1 (glucagon-like peptide) and GIP (glucose-dependent insulinotropic polypeptide) are two metabolic hormones stimulating insulin secretion via activation of their respective G protein-coupled receptors (GPCRs) (GLP-1R and GIPR). Both GLP-1R and GIPR are positively coupled to cAMP production and recruit b-arrestin2 (ARRB2).
Increasing the efficacy of GLP-1R agonists is the main focus of research in type 2 diabetes. One new strategy is to co-stimulate GLP-1R and GIPR with a dual GLP-1/GIP agonist, Tirzepatide (TZP), in order to benefit from the positive effects of both hormones on the pancreas and other target tissues.
The study, led by Dr. Magalie Ravier (Dalle-Renard’s team, IGF, Montpellier), in collaboration with Dr. Annie Varrault (Journot’s team, IGF, Montpellier), the human islet isolation platform (LTCD, IRMB, CHU Montpellier, Montpellier), and Dr. Patrick Gilon (Université Catholique de Montpellier, Montpellier), highlights the physiological role of ARRB2 in GLP-1R and GIPR signaling, and the possible functional consequences of its reduced expression in pathological situations. More specifically, this study reports that diabetogenic conditions (glucolipotoxicity) decrease ARRB2 expression in human islets. In mouse β cells, ARRB2 dampens insulin secretion by partially uncoupling cyclic AMP (cAMP)/protein kinase A (PKA) signaling at physiological doses of GLP-1, whereas at pharmacological doses, the activation of extracellular signal-related kinase (ERK)/cAMP-responsive element-binding protein (CREB) requires ARRB2. In contrast, GIP-potentiated insulin secretion needs ARRB2 in mouse and human islets. The GIPR-ARRB2 axis is not involved in cAMP/PKA or ERK signaling but does mediate GIP-induced F-actin depolymerization. Finally, the dual GLP-1/GIP agonist TZP does not require ARRB2 for the potentiation of insulin secretion.
Thus, ARRB2 plays distinct roles in regulating GLP-1R and GIPR signaling, and this study highlights (1) its role in the physiological context and the possible functional consequences of its decreased expression in pathological situations such as diabetes and (2) the importance of assessing the signaling pathways engaged by the agonists (biased/dual) for therapeutic purposes..
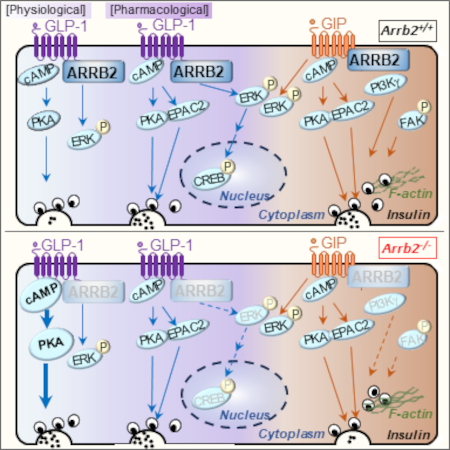
Involvement of b-arrestin2 in signalling pathways initiated by physiological and pharmacological doses of GLP-1 or in response to GIP in the pancreatic b-cell.


