MICROGLIA IN ALZHEIMER DISEASE: EACH ONE HAS ITS OWN JOB!
Microglial cells are the brain innate immune cells. Under both physiological and pathological conditions, they form a heterogeneous cell population with each subtype likely being involved in specific functions that may differentially affects brain functions.
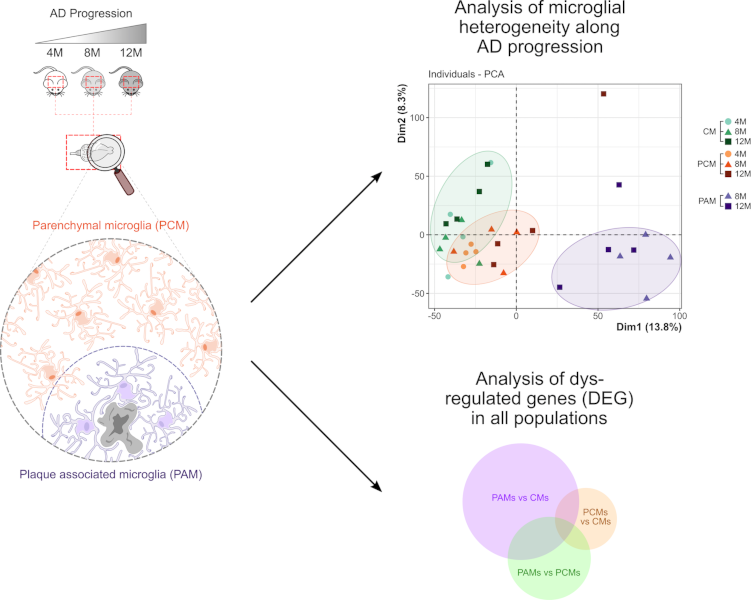
In Alzheimer’s Disease, where microglial cells are thought to play an essential role in the pathogenesis of the disease, two types of microglia can easily be distinguished. Amyloïd plaque-associated microglia (PAM) display a clearly distinct morphological phenotype compared to plaque-distant microglia (PCM), suggesting that these two microglia subtypes likely differently contribute to disease progression. A study coordinated by Hélène Hirbec in François Rassendren’s team used a spatio-transcriptomic approach to identify, without preconceived notions of the molecular and/or functional changes that would affect these cells, the genes and gene networks that are dysregulated in PAM or PCM along AD progression.
By combining laser capture microdissection and RNA-seq analysis the authors identified, in both PAM and PCM subpopulations, networks of co-deregulated genes and analyzed their potential functional roles in AD. They also investigated the dynamics of microglia transcriptomic remodeling at early, intermediate and late stages of the disease. This comprehensive study provides useful transcriptomic information regarding the respective contribution of PAM and PCM across the amyloid-ß pathology progression. In particular, it demonstrates that the proximity of microglia to amyloid-plaques dramatically alters the microglial transcriptome and reveals that these changes can have both positive and negative impacts on the surrounding cells. A local microglia heterogeneity is also demonstrated by this study. Notably, this study shed light on plaque distant microglia (PCM), which comparatively to PAM have been less well studied. The study reveals that, although the transcriptomic changes are far less striking compared to that in PAM, PCM are not bystanders of the disease. In particular, the data obtained suggest they may be involved in Aß oligomer detection and in Aß-plaque initiation, with increased contribution as the disease progresses.