Calcium is a “jack of all trades” in our cells: it converts external signals into internal messages, regulates gene expression, or activates hormone secretion. Calcium fulfills such a plurality of actions through a genuine cell code, based on the recurrence of brief calcium signals (from a hundred milliseconds to a few seconds), modulated in amplitude and/or frequency. A study coordinated by Xavier Bonnefont in the team headed by Patrice Mollard at the IGF reveals that the calcium code may encompass a wider range of timescales.
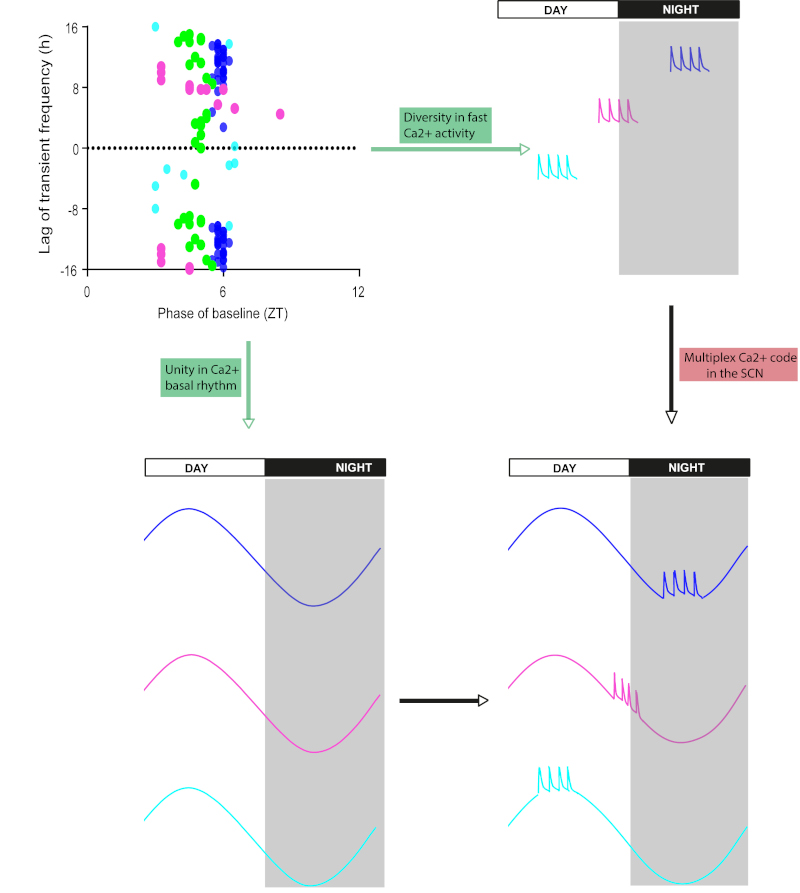
Deep in our brain, in the hypothalamus, the suprachiasmatic nuclei (SCN) host the internal clock that paces our biological rhythms in tune with the day-night cycle. SCN neurons display high calcium levels during the day and low calcium levels at night. However, investigating this ultra-slow calcium oscillation in vivo at the individual cell level has long been impossible because of the SCN location. Moreover, its relationship with typical, neuronal activity-related brief calcium signals remained unclear. To address these questions, the researchers at IGF used state-of-the-art imaging to monitor cellular calcium with a miniaturized microscope in the SCN of freely moving mice. This approach unveils that fast calcium signals in SCN neurons are small in amplitude, and superimposed upon the daily oscillation in baseline calcium concentration. Remarkably, these two calcium dynamics are independently modulated with the time of day. The baseline calcium oscillates synchronously in all recorded neurons, supporting the temporal unity that confers precision to our circadian clock. Contrastingly, fast signals underscore the functional diversity among these same neurons, some being preferentially active during the day when their calcium concentration is high, others at night when calcium level is low. Hence, nested calcium dynamics, supporting cell unity in the course of hours and cell diversity at the timescale of seconds, expand the calcium code with many new combinations in clock cells.
More generally, this work published in PNAS Nexus illustrates how calcium signaling may individualize functional cell subsets in a coherent neural network, similar to the way players in a sports team fulfill specific roles while all wearing the same team colors.