As part of the “IMPACT SANTE” call from Inserm, a funding program for groundbreaking, high-risk, and impactful health research, Cédric Leyrat, researcher in the team led by Sébastien Granier and Bernard Mouillac, is taking part in the AIR-MT project. This project, funded to the tune of 3 million Euros, is coordinated by Bruno Canard (AFMB, Marseille), in collaboration with Renaud Vincentelli (AFMB, Marseille), Olivier Schwartz (Institut Pasteur, Paris), Béatrice Nal (CIML, Marseille) and Cédric Leyrat (IGF, Montpellier).
Messenger RNAs (mRNAs) are inherently unstable molecules whose function and stability are regulated by more than 100 known post-transcriptional modifications, including RNA methylations. The past few years have seen mRNA therapeutics and vaccines revolutionize medicine, highlighting the need for precise control over synthetic mRNA stability and selective functionalization to ensure optimal cellular efficacy. The AIR-MT project aims to address this challenge by leveraging site-specific RNA methylation to enhance both mRNA stability and the balance between immune response activation and non-self protein expression.
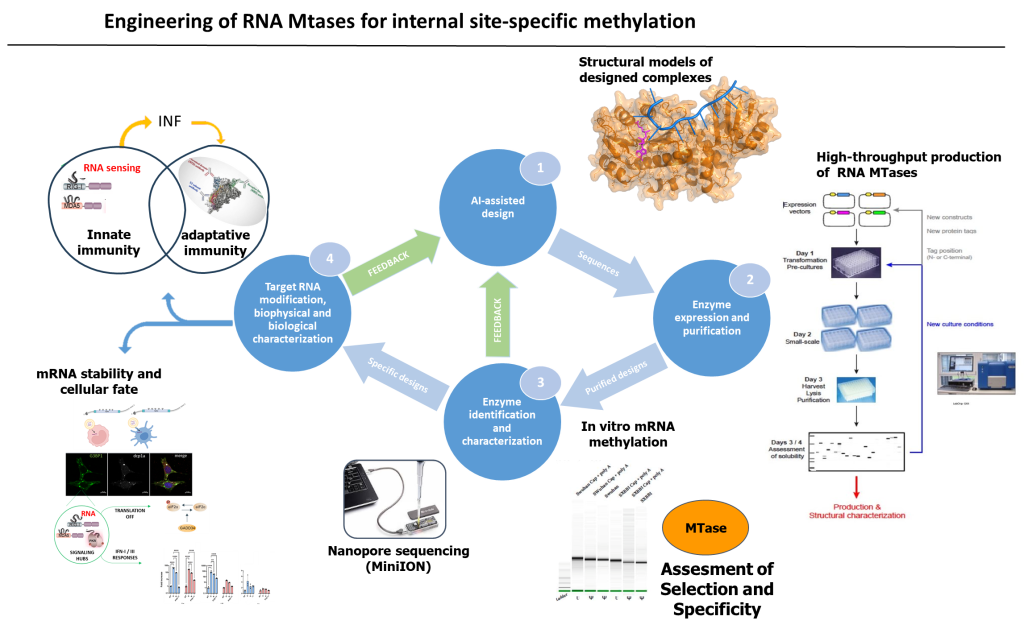
AIR-MT Project Flow. AI-assisted design generates virtual scaffolds (1). Synthetic genes are ordered, expressed, and the putative RMTases are produced, purified (2), and their specificity is analyzed using various RNA substrates. Synthetic vaccinal mRNAs are modified, and the site/atom specificity of RMTases is determined (3). These modified mRNAs are then evaluated, and the induction of the antiviral response, involving innate immunity and the immune system, is dissected (4). This constitutes a single loop in the iterative process, where promising RMTases are used to modify mRNAs with improved biophysical and biological properties. The characterization rounds performed in (3) and (4) provide feedback to step (1) (green arrows).
The key concept of AIR-MT is to develop site-specific RNA methyltransferases (RMTases) capable of modifying mRNAs to resist chemical and nuclease-mediated degradation while conferring desirable immunological properties for future mRNA-based therapeutics and vaccines. The project will harness recent breakthroughs in artificial intelligence-driven enzyme design to create synthetic RMTase folds with specific activities targeting key RNA modifications, including m6A (N6-methyladenosine), m1A (N1-methyladenosine), m5C (5-methylcytidine), and Nm (2’-O-methylation). These engineered enzymes will be screened and optimized using high-throughput methods.
To demonstrate the feasibility and impact of this approach, AIR-MT will focus on a well-defined and clinically relevant RNA target: the SARS-CoV-2 Spike mRNA, which has been instrumental in global vaccination efforts. By identifying fragile and immunogenic regions within the RNA sequence and selectively methylating them, the project aims to generate novel mRNA molecules with enhanced stability, optimized immune evasion, and improved translational efficiency. These engineered mRNAs will be evaluated in cellular and animal models, paving the way for next-generation vaccines, therapeutic RNAs, and innovative tools for biomedical research.
Beyond establishing fundamental knowledge in RNA modification and mRNA stability, AIR-MT is poised to contribute to the development of more effective and durable mRNA-based therapeutics, with broad applications in vaccine development and the treatment of various diseases.
Read the Inserm press release (in french) : https://presse.inserm.fr/impact-sante-30-millions-deuros-confies-a-linserm-pour-faire-avancer-la-recherche-en-sante/70177/


