Metabotropic glutamate receptors are key regulators of many synapses in the brain. Though for many years only eight homodimeric mGluRs were recognized, the team “Neuroreceptors, dynamics and functions” led by Philippe Rondard at the IGF in collaboration with several national and international laboratories has recently demonstrated the possible existence of 16 heterodimeric receptors, among them, the mGlu2-4 was clearly found in the brain at an even higher level than the mGlu4 homodimers (Meng et al., Nat Chem Biol 2022). Surprisingly, while both mGlu2 and mGlu4 homodimers can activate G proteins, mGlu4 is the signaling subunit in the mglu2-4 heterodimer. Thanks to an international collaboration involving Jianfeng Liu’s lab in Wuhan and Yan Zhang’s lab in HangZhou (China), the teams “Integrative biophysics of membranes” led by Emmanuel Margeat at CBS and the “Neuroreceptors, dynamics and functions” led by Philippe Rondard at IGF in Montpellier have now reported on the structural basis of such an oriented asymmetry of the mGlu2-4 heterodimer activation. The authors show that both subunits do not undergo similar conformational changes explaining the oriented asymmetry. The work also provides the structure of the heterodimer in intermediate states, with only one subunit activated, and provides an explanation for the partial activity of these states using single molecule FRET.
This work was accepted for publication in Nature Communications.
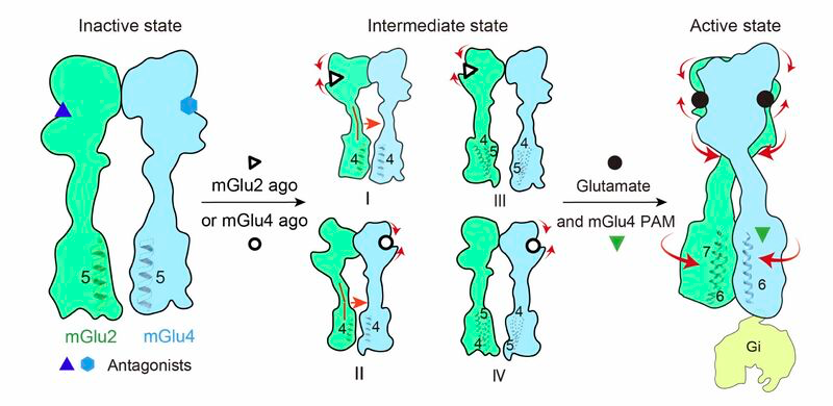
Schematic of the sequential conformational changes for mGlu2-4 heterodimer during activation. Cartoon illustrates activation-related transitions including inter-subunit and intra-subunit from the inactive, through one agonist-bound intermediate state to the agonist, PAM and G protein-bound full active state.


