TUFT CELLS MEDIATE PANETH CELL-DRIVEN GUT DYSBIOSIS AND INFLAMMATION
The intestinal epithelium represents the largest exchange area with the external environment and constitutes a crucial barrier to protect the organism against pathogens and infectious agents found in the intestinal lumen. The intestinal microbiota plays a major role in this function, and alterations in its composition (dysbiosis) actively participate to the development and severity of multiple diseases such as inflammatory bowel disease (IBD), metabolic or psychiatric disorders…
The team headed by Philippe Jay at the IGF identified a novel multi-step mechanism for dysbiosis initiation via a cross talk between two epithelial cell types: Paneth cells, that control microbiota by secreting antimicrobial peptides, and tuft cells, key epithelial sentinels that regulate mucosal immunity in case of parasitic infection. Initial alterations in Paneth cell, as observed in IBD and obese patients, induce mild remodeling of microbiota, with amplification of succinate-producing species. The increase in succinate, normally low in physiological conditions, is a warning signal, which has the ability to specifically activate tuft cells through the succinate receptor. Tuft cells then induce a type 2 immune response that, in turn, exacerbates the Paneth cell defaults, thus promoting dysbiosis and chronic inflammation.
This study reveals a novel function of tuft cells in promoting dysbiosis following Paneth cell dysfunction, and highlights an unknown role of Paneth cells in controlling a balanced microbiota to prevent an inadequate and permanent tuft cell activation. Our findings open perspectives for therapies targeting tuft cell activity for a more effective management of dysbiosis-associated chronic diseases
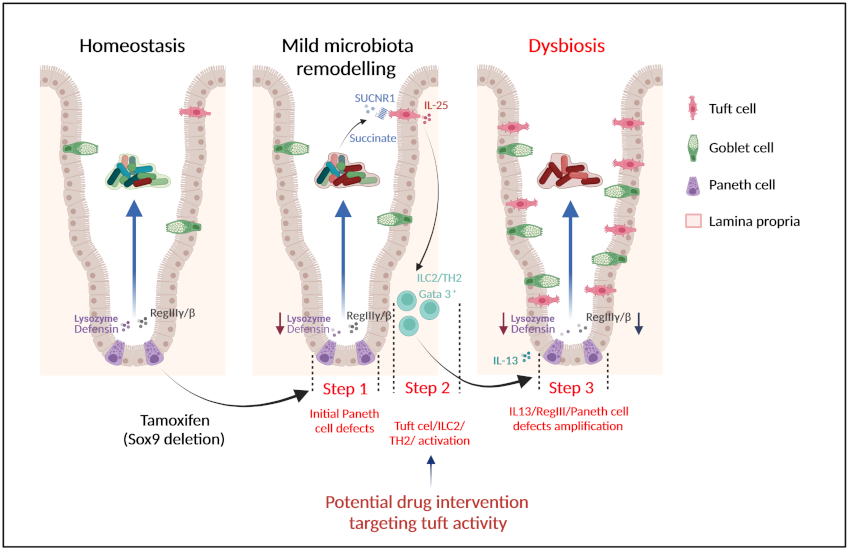
Multi-step mechanism for establishment of dysbiosis and inflammation due to a Paneth cells/succinate/tuft cells/ immune cells circuit.


