Cell-to-cell and cell-environment communication typically occurs through small molecules, with signal transduction into cells mediated by proteins known as receptors. In the mammalian nervous system, L-glutamate serves as the primary excitatory neurotransmitter, playing key roles in synaptic plasticity and physiological processes such as learning and memory. L-glutamate binds to ionotropic glutamate receptors, which function as ligand-gated ion channels (e.g., the N-methyl-D-aspartate (NMDA) receptor), and to metabotropic glutamate receptors (mGlu), which belong to the G protein-coupled receptor (GPCR) family. Aberrant glutamatergic transmission is associated with numerous neurological disorders, rendering these receptors significant therapeutic targets.
In addition to glutamate, the orthosteric agonist, mGlu receptors can be modulated allosterically by molecules categorized as negative or positive allosteric modulators (NAM / PAM). Among the eight members of the mGlu receptor family, mGlu5 plays an essential role in synaptic plasticity, and its dysfunction has been linked to neurological disorders such as autism, Parkinson’s and Alzheimer’s diseases, making mGlu5 a validated drug target. More specifically, positive allosteric modulation of mGlu5 holds therapeutic potential for mitigating symptoms of schizophrenia. Despite its promise, designing specific PAMs for individual mGlu isoforms remains a challenging task.
The architecture of the mGlu receptor comprises an extracellular Venus-Fly Trap (VFT) domain connected to the transmembrane domain (7TM) via a cysteine-rich domain (CRD). When L-glutamate binds to the receptor’s VFTs, it induces VFTs closure, triggering a substantial conformational change conveyed to the 7TM domain via the CRD. This transition stabilizes an active state, facilitating optimal recruitment and binding of intracellular signalling partners such as G proteins. PAMs bind to the 7TM domain approximately 100 Å from the L-glutamate binding site, enhancing receptor signalling. The combination of multiple ligands binding to the receptor can result in distinct signalling outcomes, a phenomenon known as biased signalling, which elicits diverse cellular responses. Understanding how PAMs bind and influence the dynamics of mGlu receptor activation is essential for designing novel therapeutic molecules.
Using cryo-EM, the team “Molecular and Structural Mechanisms of Neuromodulation” headed by par Guillaume Lebon et Cyril Goudet determined the structures of detergent-purified full-length mGlu5 bound to the high-affinity orthosteric agonist quisqualate (an L-glutamate analogue) and three PAMs with distinct chemistry and pharmacology. These structures revealed differential PAM binding modes and their effects on receptor conformation, highlighting key residues involved in modulating receptor activity. The three PAMs occupy a similar binding pocket in the 7TM domain but exhibit slight variations in their interactions, as confirmed through pharmacological characterization. These residues, previously implicated in stabilizing the inactive conformation of the 7TM domain, appear to interact differently with each PAM. Notably, PAMs and NAMs share a common binding site in mGlu5, with subtle differences influencing receptor activation.
This study highlights the diversity of PAM binding modes and their effects on stabilizing the 7TM interface, a critical step for receptor activation and signalling. The findings also provide a structural framework for the development of structure-based drug discovery programs targeting mGlu5 PAMs.
This work was accepted for publication in Nature Communications.
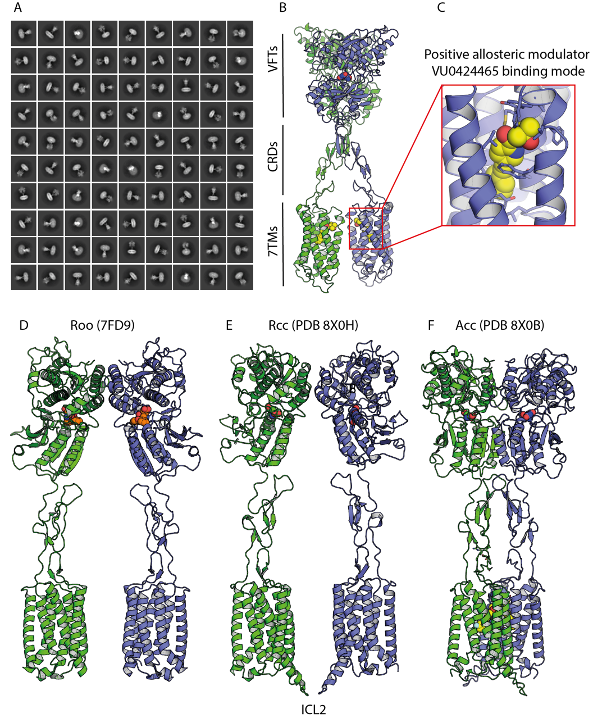
Single-particle cryo-EM analysis of the agonist- and PAM-bound mGlu5 receptor in detergent micelles. a) Averaged mGlu5 receptor particles obtained after 2D classification of PAM-bound mGlu5 receptors, computed from high-resolution cryo-EM images. b) 3D model of the mGlu5 receptor bound to the PAM VU0424465, showing one PAM molecule bound to each 7TM domain. The inset provides a zoomed-in view of the VU0424465 binding pocket, highlighting key interaction residues. c) Inactive state (Roo), where the antagonist LY1341494 is bound to the open VFT domain (PDB 7FD9). d) Intermediate activation state (Rcc) identified in this study, in which quisqualate is bound to closed VFT domains but no PAM is present. The 7TM domains remain far apart, similar to the inactive Roo state. e) Active state (Acc), showing the receptor bound to quisqualate in the close VFTs and VU0424465 in the 7TM domains. In the graphical representation of the mGlu5 receptor dimer, protomers are coloured green and blue, while ligands are depicted in sphere representation: LY341495 (orange), quisqualate (green and blue), and VU0424465 (yellow).


